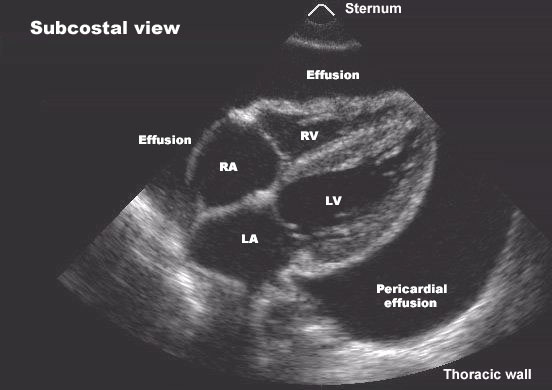
Takotsubo Cardiomyopathy (Takotsubo Syndrome, Stress Cardiomyopathy)
Definition
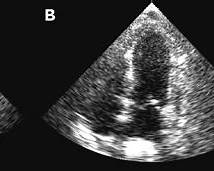
Acute, usually reversible syndrome of transient LV (± RV) systolic dysfunction that often mimics ACS (chest pain, ECG changes, troponin rise) but typically occurs without culprit obstructive coronary occlusion; commonly triggered by emotional/physical stress. ([NCBI][1])
Pathophysiology
- Catecholamine surge → myocardial stunning, microvascular dysfunction, coronary spasm, direct myocyte injury.
- Regional wall-motion abnormality extends beyond a single epicardial coronary territory (typical pattern).
- Variants: apical ballooning (classic), mid-ventricular, basal/reverse, focal, sometimes biventricular involvement. ([Radiopaedia][2])
Causes / Triggers
- Emotional: grief, fear, anger
- Physical: sepsis, stroke/SAH, surgery, asthma/COPD exacerbation, trauma, pain
- Drugs: catecholamines, sympathomimetics (risk/association)
Clinical features
- Chest pain, dyspnea, syncope
- May present with:
* Acute HF/pulmonary edema
* Cardiogenic shock
* Arrhythmias (AF, VT/VF), QT prolongation → torsades
* Mechanical complications (rare): LV rupture, acute MR
Investigations / Diagnosis (stepwise)
- ECG: ST elevation or depression, T-wave inversion, QT prolongation (often dynamic).
- Biomarkers: Troponin modestly elevated; BNP/NT-proBNP often high.
- Echocardiography: regional LV dysfunction pattern; assess LVOT obstruction and MR.
- Coronary angiography (often required initially to exclude ACS when STEMI-like): frequently non-obstructive or non-culprit CAD. ([NCBI][1])
- Cardiac MRI: helpful to exclude myocarditis/infarction; look for edema and late gadolinium enhancement patterns (typically absent or limited vs MI).
- Consider diagnostic criteria frameworks (e.g., InterTAK / Mayo-style concepts). ([Radiopaedia][3])
Key differentials
- ACS (STEMI/NSTEMI)
- Myocarditis (viral/immune checkpoint inhibitor)
- MINOCA
- Coronary spasm, microvascular angina
- Pulmonary embolism, aortic dissection (if presentation atypical)
Management (practical, stepwise)
A. Initial ED/ACS-mimic phase (until ACS excluded)
- Treat as ACS: aspirin, anticoagulation, nitrates/analgesia as appropriate until angiography/diagnostic clarity (local protocol).
B. After Takotsubo likely/confirmed
- Supportive HF therapy (if congested):
* Oxygen/ventilatory support, diuretics if volume overloaded.
- Hemodynamic phenotype matters
* If cardiogenic shock WITHOUT LVOT obstruction: consider vasopressors (e.g., norepinephrine) ± mechanical circulatory support.
* If LVOT obstruction present: avoid inotropes (may worsen obstruction); use cautious fluids (if not congested), short-acting beta-blocker, and vasoconstrictor if needed.
- Thromboembolism prevention
* If severe LV dysfunction and/or LV apical thrombus: anticoagulate (often for ~3 months or until LV recovery; individualized).
C. Arrhythmias
- Correct electrolytes (K, Mg), avoid QT-prolonging drugs, monitor for torsades/VT.
D. Follow-up
- Repeat echo (often 4–12 weeks) to document recovery.
- Address triggers, anxiety/depression, cardiac rehab where appropriate.
Drug details (common choices)
1) Beta-blocker (e.g., Metoprolol)
- Indications: rate control, LVOT obstruction management (short-acting agents often used acutely), possibly reduce adrenergic effect (evidence mixed).
- Mechanism: β1 blockade → ↓HR/contractility; reduces LVOT gradient.
- Adult dosing:
* Metoprolol tartrate: 25–50 mg PO q6–12h (titrate); IV: 5 mg slow IV q5 min up to 15 mg if needed (monitor BP/HR).
- Paeds (specialist): 1–2 mg/kg/day PO divided (varies by age/indication).
- PK: hepatic (CYP2D6), half-life ~3–7 h (longer in poor metabolizers).
- Adverse: bradycardia, hypotension, bronchospasm (less with β1 selective), fatigue, masking hypoglycemia.
- Contra: severe bradycardia, 2nd/3rd degree AV block, cardiogenic shock (unless specific LVOT strategy), severe asthma (relative).
- Interactions: other AV nodal blockers (verapamil/diltiazem), digoxin; CYP2D6 inhibitors (fluoxetine, paroxetine) ↑levels.
- Monitoring: HR, BP, wheeze, ECG PR interval.
- Counselling: don’t stop abruptly; report dizziness, wheeze.
2) ACE inhibitor (e.g., Enalapril) / ARB (e.g., Losartan)
- Indications: LV systolic dysfunction during recovery phase.
- Mechanism: RAAS blockade → ↓afterload, remodeling benefit in HFrEF physiology.
- Adult dosing:
* Enalapril 2.5 mg PO BID → titrate (target often 10–20 mg BID in HFrEF).
* Losartan 25–50 mg daily → titrate (up to 100 mg daily).
- Paeds (specialist):
* Enalapril ~0.08–0.6 mg/kg/day divided.
- PK: enalapril prodrug → enalaprilat; renal elimination.
- Adverse: hypotension, AKI, hyperkalemia, cough (ACEi), angioedema.
- Contra: pregnancy, bilateral renal artery stenosis, history angioedema (ACEi).
- Interactions: K supplements/spironolactone ↑K, NSAIDs ↑AKI risk, lithium ↑levels.
- Monitoring: creatinine, potassium, BP.
- Counselling: hydrate; avoid NSAID overuse; seek care for facial swelling.
3) Loop diuretic (Furosemide)
- Indications: pulmonary edema/congestion.
- Mechanism: inhibits NKCC2 in loop → natriuresis.
- Adult dosing: 20–40 mg IV/PO; titrate to diuresis.
- Paeds: 0.5–1 mg/kg/dose IV/PO.
- Adverse: hypokalemia, hyponatremia, ototoxicity (high IV dose), dehydration.
- Monitoring: weight, urine output, K/Mg/Cr.
- Counselling: morning dosing, cramps → check electrolytes.
4) Anticoagulation (example: Apixaban / Warfarin)
- Indications: LV thrombus or high-risk severe apical akinesis.
- Mechanism: DOAC Xa inhibition (apixaban) / vitamin K antagonism (warfarin).
- Adult dosing: Apixaban 5 mg BID (dose-reduce if criteria met); Warfarin to INR 2–3.
- Paeds: specialist only.
- Adverse: bleeding; warfarin—skin necrosis (rare), teratogenic.
- Interactions: DOAC—strong CYP3A4/P-gp inhibitors/inducers; warfarin—many drug/food interactions.
- Monitoring: bleeding signs; warfarin INR.
Brugada Syndrome
Definition
An inherited primary arrhythmia syndrome (channelopathy) characterized by a type 1 Brugada ECG pattern (coved ST elevation in right precordial leads) and risk of polymorphic VT/VF and sudden cardiac death, often in structurally normal hearts. ([AHA Journals][4])
Pathophysiology
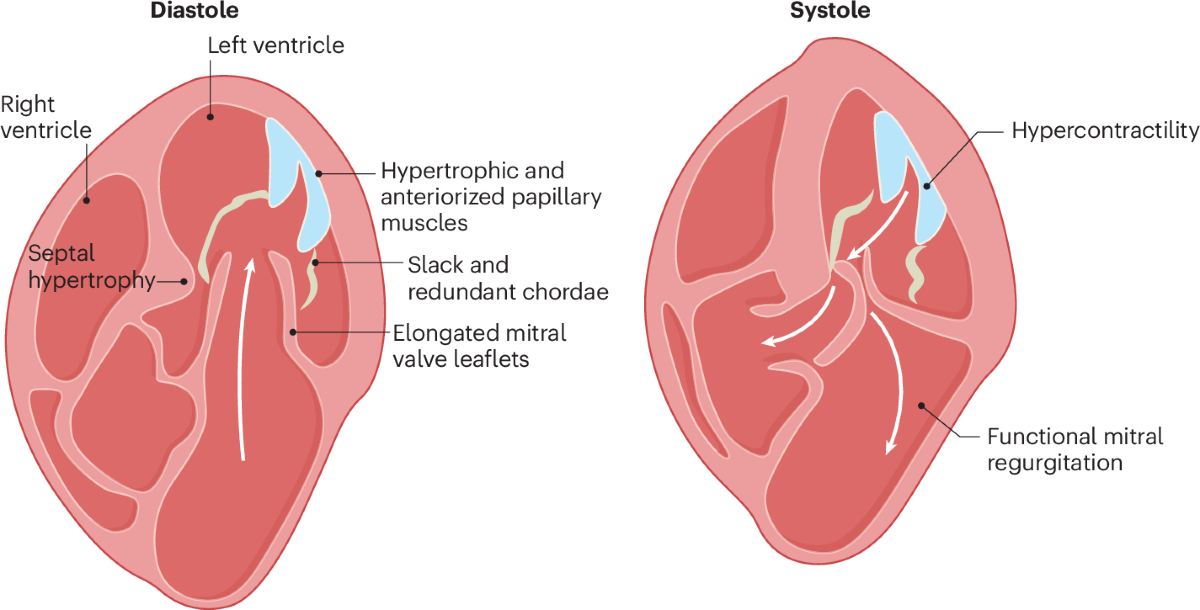
- Most commonly involves reduced cardiac sodium current (e.g., SCN5A variants) → conduction/repolarization abnormalities in RV outflow tract region → phase 2 reentry and VF susceptibility. ([AHA Journals][4])
Triggers / Precipitants
- Fever (major), alcohol binge
- Large meals, vagal predominance (rest/sleep)
- Sodium-channel blocking drugs, some anesthetics/psychotropics
- Electrolyte disturbances (hyperkalemia), cocaine
Clinical features
- Syncope (often nocturnal), palpitations
- Seizure-like activity (from cerebral hypoperfusion)
- Agonal respirations at night
- Family history of unexplained SCD (often young)
Diagnosis
ECG patterns
- Type 1 (diagnostic): coved ST elevation ≥2 mm in ≥1 of V1–V2 (placed in 2nd–4th intercostal spaces) with negative T wave.
- Type 2/3 are not diagnostic alone.
Drug challenge (unmasking type 1)
- Ajmaline / flecainide / procainamide in controlled setting with resuscitation ready (specialist EP).
Exclude phenocopies
- Ischemia, myocarditis, PE, hyperkalemia, hypothermia, pectus excavatum, ARVC, etc.
Differential diagnoses
- ARVC (especially RVOT involvement)
- Early repolarization syndrome
- RBBB variants
- Acute pericarditis (ST elevation pattern differs)
- STEMI in anterior leads (clinical context)
Risk stratification (high-yield)
Higher risk if:
- Prior cardiac arrest/VF
- Arrhythmic syncope
- Spontaneous type 1 pattern
- Inducible VT/VF on EP study (controversial, adjunct)
- Family history alone is not sufficient for ICD without other factors.
Management (stepwise)
1) Universal measures
- Aggressively treat fever (antipyretics + evaluation) to reduce VF risk.
- Avoid contraindicated/proarrhythmic drugs (see BrugadaDrugs lists). ([Brugada Drugs][5])
- Avoid excess alcohol, dehydration; correct electrolytes.
2) ICD
- Classically indicated after aborted SCD or documented VT/VF.
- Consider in arrhythmic syncope with spontaneous type 1 pattern (specialist risk assessment).
3) Electrical storm / recurrent VF
- Isoproterenol infusion (acute) and/or quinidine (acute + chronic) are commonly used strategies; manage in ICU with EP input. ([OUP Academic][6])
- Catheter ablation of RVOT substrate (selected refractory cases).
Drug details (Brugada-focused)
A) Isoproterenol (Isoprenaline) – acute electrical storm
- Indication: VF storm in Brugada; suppresses VF by increasing inward calcium current and heart rate (reduces ST elevation/arrhythmogenic substrate).
- Mechanism: non-selective β agonist → ↑HR, ↑AV conduction, ↑cAMP.
- Adult dosing (ICU): start 0.02–0.1 μg/kg/min IV infusion, titrate to suppress VF and raise HR (institution protocols vary).
- Paeds: specialist dosing; titrated infusion.
- PK: very short acting; IV continuous.
- Adverse: tachyarrhythmias, ischemia, tremor, hypotension.
- Contra/caution: active ischemia, severe aortic stenosis, uncontrolled tachyarrhythmias.
- Monitoring: continuous ECG, BP, ischemia signs, K/Mg.
B) Quinidine – prevention of VF / adjunct
- Indications: recurrent VT/VF, ICD shocks, bridging/when ICD not feasible.
- Mechanism: class Ia Na+ block + IKr block; also reduces Ito current effect in RV epicardium (anti-VF in Brugada rationale).
- Adult dosing (typical):
* Quinidine sulfate 200–400 mg PO every 6–8 h (or extended-release regimens depending availability); titrate with ECG/QT monitoring.
- Paeds: specialist only.
- PK: hepatic metabolism; variable formulations.
- Adverse: QT prolongation → torsades, diarrhea, cinchonism (tinnitus/headache), thrombocytopenia (rare), hypoglycemia.
- Contra: prolonged QT/torsades history, myasthenia gravis (worsening), significant AV block without pacing.
- Interactions: other QT-prolongers, digoxin (↑levels), warfarin (↑INR), CYP/P-gp interactions.
- Monitoring: baseline and follow-up ECG (QTc), electrolytes, symptoms.
C) Antipyretics (Paracetamol/Acetaminophen)
- Indication: fever control (risk-reduction measure).
- Adult dosing: 500–1000 mg PO q6–8h (max per local guidance; often 3–4 g/day depending risk).
- Paeds: 10–15 mg/kg/dose q4–6h (max daily per age/weight guidelines).
- Adverse: hepatotoxicity in overdose.
- Counselling: avoid duplicate combo cold meds.
Sudden Cardiac Death (SCD) / Sudden Cardiac Arrest (SCA)
Definition
- SCA: abrupt loss of cardiac mechanical function leading to collapse (often VF/VT, PEA, asystole).
- SCD: unexpected death due to cardiac causes, often within a short time of symptom onset (definitions vary by system). ([Radiopaedia][7])
Causes (organized)
1) Structural heart disease (commonest overall)
- Coronary artery disease / acute MI scar-related VT
- Cardiomyopathies: HCM, DCM, ARVC, myocarditis, sarcoidosis
- Valvular (severe AS), congenital anomalies
2) Primary electrical diseases
- Brugada, Long QT, Short QT, CPVT, early repolarization syndrome
3) Non-cardiac mimics to consider
- Massive PE, intracranial bleed, severe hemorrhage, overdose, hypoxia
Warning symptoms (when present)
- Exertional syncope, unexplained syncope
- Palpitations with presyncope
- Family history of SCD <50 years
- Known cardiomyopathy/low EF, prior MI scar
Immediate management of SCA (adult, high-yield)
- Early CPR + early defibrillation are the biggest survival determinants in VF/pVT. ([AHA Journals][8])
- Follow AHA ALS algorithms (shockable vs non-shockable). ([cpr.heart.org][9])
ACLS drug details (common)
1) Epinephrine (Adrenaline)
- Indication: cardiac arrest (VF/pVT after shocks + CPR; PEA/asystole early).
- Mechanism: α1 vasoconstriction → ↑coronary/cerebral perfusion; β effects.
- Adult dosing: 1 mg IV/IO every 3–5 min during arrest.
- Paeds: 0.01 mg/kg IV/IO (0.1 mL/kg of 0.1 mg/mL) q3–5 min.
- Adverse: post-ROSC tachyarrhythmias, hypertension, ischemia.
- Monitoring: rhythm, ETCO₂, perfusion after ROSC.
2) Amiodarone
- Indication: refractory VF/pVT.
- Mechanism: class III predominant (K+ block) + multi-channel effects.
- Adult dosing: 300 mg IV/IO bolus, may give additional 150 mg.
- Paeds: 5 mg/kg IV/IO bolus, may repeat up to max per protocol.
- PK: very long half-life; tissue accumulation.
- Adverse: hypotension (IV solvent-related), bradycardia; chronic toxicities if continued.
- Interactions: warfarin, digoxin (↑levels), QT-prolongers.
- Monitoring: ECG/QT, BP.
3) Lidocaine (alternative to amiodarone)
- Adult dosing: 1–1.5 mg/kg IV/IO, then 0.5–0.75 mg/kg (max per protocol).
- Adverse: CNS toxicity, seizures (esp. high dose).
4) Magnesium sulfate
- Indication: torsades de pointes / suspected hypomagnesemia.
- Adult dosing: 1–2 g IV/IO.
- Adverse: hypotension with rapid infusion.
Post–cardiac arrest care (essentials)
- Treat cause (ACS/PE/etc.), optimize oxygenation/ventilation, manage temperature, hemodynamics, neurologic prognostication (protocolized ICU care). ([cpr.heart.org][9])
Secondary prevention (after survival / high-risk identification)
- ICD for many patients with:
* Prior VF/VT arrest not due to reversible cause
* Cardiomyopathy with persistently low LVEF per guideline thresholds (specialist-based).
- Targeted therapy for cause:
* Revascularization for ischemia
* HF guideline-directed therapy
* Ablation for recurrent VT
* Channelopathy-specific measures (e.g., Brugada fever/drug avoidance; LQTS beta-blocker; CPVT beta-blocker + flecainide)
Screening / workup after unexplained SCA or family SCD
- ECG (including high right precordial leads), echo, coronary imaging as indicated, CMR, exercise testing, Holter/loop, genetic counseling/testing when inherited syndrome suspected. ([HRS][10])