1) Short definition
Metabolic syndrome is a clustering of interrelated metabolic risk factors (central obesity, dyslipidaemia, raised blood pressure, and hyperglycaemia/insulin resistance) that together markedly increase the risk of type 2 diabetes mellitus (T2DM), atherosclerotic cardiovascular disease (ASCVD), and all-cause mortality. ([PMC][1])
2) Pathophysiology — how it develops
- Central (visceral) adiposity is the usual initiating factor: adipose tissue dysfunction → increased free fatty acids, adipokine imbalance (↓adiponectin, ↑leptin resist., proinflammatory cytokines) → systemic inflammation and insulin resistance.
- Insulin resistance in muscle/liver/adipose leads to hyperinsulinaemia, impaired glucose regulation, and dyslipidaemia (↑TG, ↓HDL, small dense LDL).
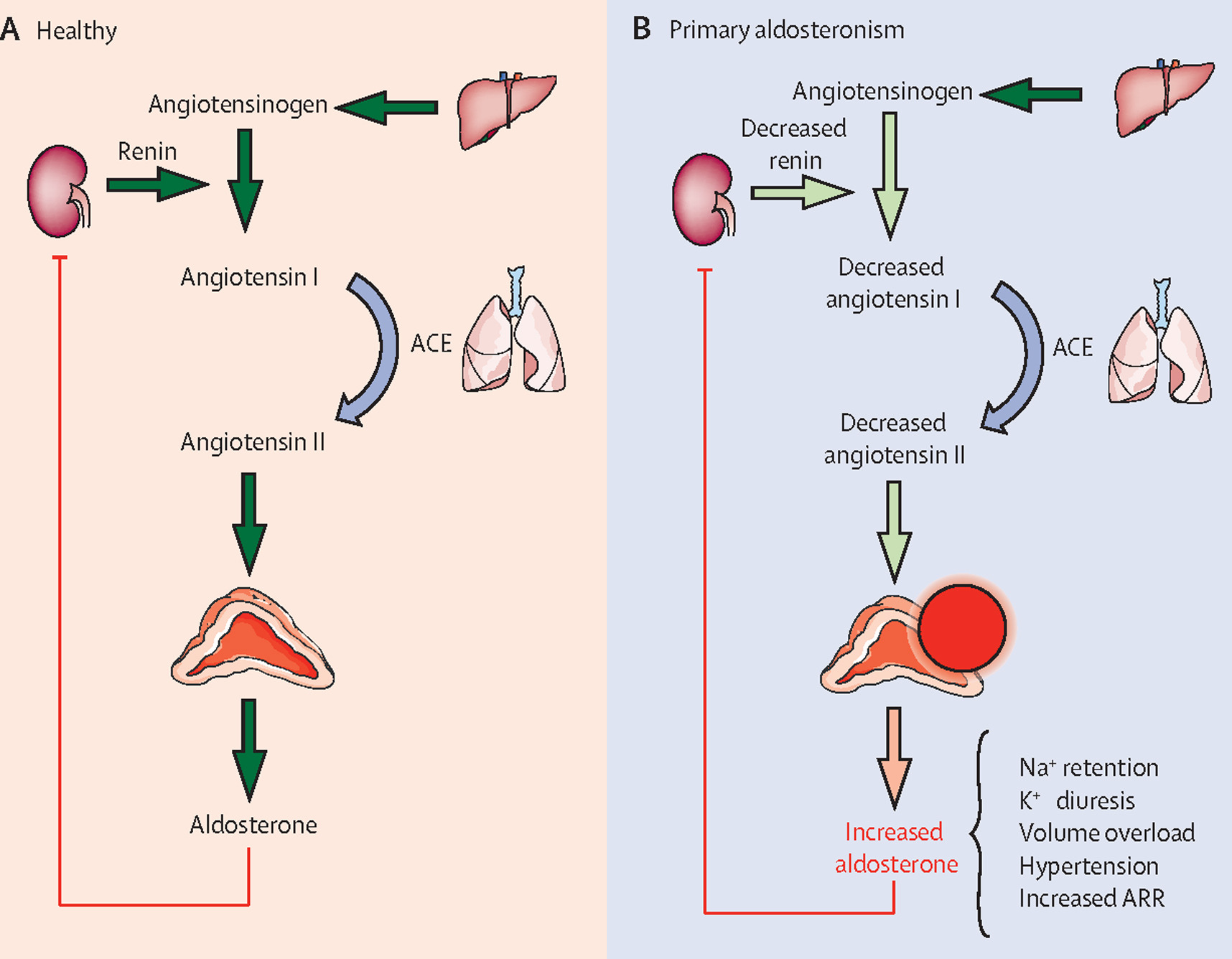
- Hypertension arises from sympathetic activation, RAAS alterations, sodium retention, and endothelial dysfunction.
- Combined, these produce accelerated atherogenesis and increased diabetes risk. ([PMC][2])
3) Major causes & risk factors
- Obesity (especially central/abdominal) — strongest modifiable risk.
- Physical inactivity.
- Unhealthy diet (high refined carbs, trans fats, excess calories).
- Genetic predisposition and family history of T2DM/obesity.
- Age (risk rises with age), male sex (varies), ethnicity (South Asian, Hispanic, Middle Eastern higher risk per same BMI).
- Smoking, excessive alcohol, certain medications (antipsychotics, some glucocorticoids). ([PMC][3])
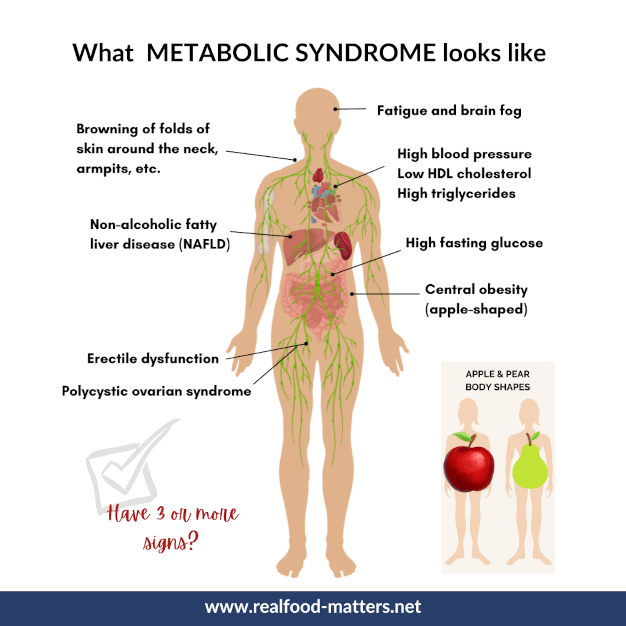
4) Clinical features (what you see / ask for)
- Truncal obesity (increased waist circumference).
- May be asymptomatic; often identified by screening tests.
- Associated findings: acanthosis nigricans (marker of insulin resistance), elevated blood pressure, xanthelasma or tendon xanthomas (if dyslipidaemia severe).
- Often coexists with nonalcoholic fatty liver disease (NAFLD). ([PMC][4])
5) Diagnostic criteria — commonly used definitions (choose one; ATP III and IDF are most used)
A. NCEP ATP III (Adult Treatment Panel III; commonly used) — diagnosis = any 3 of 5:
- Waist circumference: >102 cm (men) or >88 cm (women).
- Triglycerides ≥150 mg/dL (1.7 mmol/L) or treatment for elevated TG.
- HDL-C <40 mg/dL (men) or <50 mg/dL (women) or on treatment.
- Blood pressure ≥130/85 mm Hg or receiving antihypertensive treatment.
- Fasting plasma glucose ≥100 mg/dL (5.6 mmol/L) or on treatment for hyperglycaemia. ([PMC][1])
B. IDF (International Diabetes Federation) — central obesity (ethnicity-specific waist cutoff; e.g., ≥90 cm men / ≥80 cm women for South Asians) required, plus any two of the remaining four (TG, HDL, BP, fasting glucose). Use this for population screening where ethnicity matters. ([International Diabetes Federation][5])
C. WHO / EGIR / others — older definitions emphasize insulin resistance or glycaemic abnormalities; less used for routine screening. ([Iris][6])
6) Key investigations (baseline and follow-up)
- Anthropometry: waist circumference, BMI.
- BP measurement: seated, averaged.
- Fasting labs: fasting plasma glucose (or HbA1c), lipid profile (TG, HDL, LDL), liver enzymes (ALT/AST) for NAFLD screening.
- Optional/if indicated: fasting insulin (research), oral glucose tolerance test (OGTT) if IFG/IGT suspected, urine albumin creatinine ratio, ECG (if ASCVD risk high).
- Repeat monitoring: weight/waist and BP frequently (every visit), fasting glucose/HbA1c and lipids per guideline intervals (e.g., 3–12 months depending on control). ([Ahad Journals][7])
7) Differential diagnoses / mimics
- Cushing syndrome (central obesity with hypertension, glucose intolerance).
- Hypothyroidism (weight gain, hyperlipidaemia).
- Familial combined hyperlipidaemia or familial dyslipidaemias (look for family pattern, tendon xanthomas).
- Secondary causes of hypertension (renal disease, endocrine causes).
Evaluate clinical clues and targeted labs if suspected.
8) Management — overall approach (stepwise)
A — OVERARCHING PRINCIPLE
Treat risk factors and root causes. Lifestyle modification is first-line for almost everyone; pharmacotherapy targets individual components (hyperglycaemia, dyslipidaemia, hypertension) and, when appropriate, weight-loss agents or bariatric surgery for severe obesity. Shared decision making and long-term follow-up are essential. ([ScienceDirect][8])
B — Non-pharmacologic (cornerstone)
- Weight loss: aim for ≥5–10% body weight loss (improves insulin sensitivity, BP, lipids).
- Dietary changes: Mediterranean-style diet, reduced refined carbs/sugary drinks, increase whole grains, fibre, lean proteins; caloric deficit for weight loss. Time-restricted eating / intermittent fasting has emerging supportive data for metabolic improvements (discuss with patient). ([EatingWell][9])
- Physical activity: ≥150 min/week moderate aerobic + ≥2 sessions/week strength training.
- Smoking cessation and limit alcohol.
- Sleep optimization and stress reduction.
- Address comorbidities: obstructive sleep apnoea, depression, NAFLD. ([ScienceDirect][8])
C — Pharmacologic (principles)
Treat each component guided by existing disease-specific guidelines (e.g., antihypertensive treatment per BP guidelines, statin therapy based on ASCVD risk). Consider agents that address multiple problems (e.g., GLP-1 receptor agonists for weight and hyperglycaemia). Below are commonly used drug classes and detailed drug notes. ([MDPI][10])
D — Advanced/other options
- GLP-1 receptor agonists (semaglutide, liraglutide) and SGLT2 inhibitors have beneficial metabolic/Cardio-renal profiles in T2DM and obesity—use per indication and specialist guidance.
- Bariatric surgery for BMI ≥40 or ≥35 with comorbidities when medical therapy fails — produces large improvements in metabolic syndrome components. ([MDPI][10])
9) When to start aspirin or more aggressive preventive therapy
Aspirin for primary prevention is not routinely recommended; consider only when ASCVD risk/benefit favours use (individualized assessment). Use statins when indicated by ASCVD risk or LDL thresholds — statins are cornerstone for ASCVD risk reduction. ([Ahad Journals][7])
10) Prognosis
Without intervention, metabolic syndrome substantially increases 5–10-year risk of T2DM and ASCVD. Effective lifestyle change and control of components reduce risk; some interventions (weight loss, GLP-1 RAs, bariatric surgery) can even cause remission of T2DM in selected patients. ([PMC][4])
11) Practical follow-up schedule (example)
- 1–3 months after major lifestyle change: weight, waist, BP, fasting glucose/HbA1c, lipids if started therapy.
- Thereafter 3–12 months depending on stability and treatments.
- Annual comprehensive cardiovascular risk review.
12) Authoritative external links (key reading)
- NCEP ATP III / Circulation (Grundy et al.): definition and guidance. ([Ahad Journals][7])
- International Diabetes Federation (IDF) — metabolic syndrome definition (ethnicity cutoffs PDF). ([International Diabetes Federation][5])
- WHO 1999 report (original working definition). ([Iris][6])
- Review articles on changing definitions and management. ([PMC][2])
- Recent lifestyle trials and systematic evidence (JAMA, 2024–2025). ([JAMA Network][11])
(Clicking the citations will open the source pages.)
13) Drug summaries — details you requested (indication, MOA, usual dosing, PK summary, major adverse effects, contraindications, key interactions, monitoring, patient counselling)
> Note: dosing below is for typical adult use. Pediatric use varies by drug — metabolic syndrome management focuses on adults; mention pediatric dosing only if indicated by a specialist.
1) Metformin
- Indication: First-line for impaired fasting glucose / T2DM; frequently used in insulin resistance / prediabetes in overweight patients.
- Mechanism: Decreases hepatic gluconeogenesis, increases peripheral insulin sensitivity.
- Usual adult dosing: Start 500 mg once daily or 500 mg twice daily with meals; titrate weekly as tolerated to 2000 mg/day (max 2550 mg/day immediate-release depending on regimen). Extended-release alternatives exist (e.g., 500–2000 mg/day).
- PK: Oral absorption; bioavailability ~50–60%; not metabolized; excreted unchanged by kidney (t½ ~6 hours).
- Common adverse effects: GI upset (nausea, diarrhea), metallic taste. Rare but serious: lactic acidosis (very rare; risk ↑ with severe renal impairment).
- Contraindications / cautions: eGFR <30 mL/min/1.73 m² (contraindicated); caution if eGFR 30–45—dose reduction and specialist guidance. Avoid with acute severe illness, hypoxia, significant hepatic impairment.
- Interactions: Contrast-related acute kidney injury risk—hold per local protocols for iodinated contrast if eGFR low. Drugs that alter renal function (ACEi/ARBs, diuretics, NSAIDs) require monitoring.
- Monitoring: Baseline and periodic renal function (eGFR), B12 levels if long-term (metformin can reduce B12). Monitor glucose/HbA1c.
- Counselling: Take with food to reduce GI effects; report unexplained myalgia, hyperventilation, severe lethargy (rare lactic acidosis signs). ([MDPI][10])
2) Atorvastatin (example high-intensity statin)
- Indication: LDL-lowering for primary/secondary prevention of ASCVD; metabolic syndrome patients often meet criteria if elevated ASCVD risk.
- Mechanism: HMG-CoA reductase inhibitor → reduces LDL synthesis, increases LDL receptor expression.
- Usual adult dosing: 10–80 mg once daily; high-intensity typically 40–80 mg. Choose dose per ASCVD risk and LDL goal.
- PK: Oral; high bioavailability; metabolized CYP3A4; t½ ~14 hours.
- Adverse effects: Myalgias, elevated liver enzymes; rare rhabdomyolysis.
- Contraindications: Active liver disease; pregnancy/breastfeeding.
- Interactions: CYP3A4 inhibitors (clarithromycin, some antifungals) increase statin levels → ↑myopathy risk. Avoid with strong inhibitors or reduce dose. Fibrate co-use increases myopathy risk (be cautious; fenofibrate is safer than gemfibrozil if combined under specialist advice).
- Monitoring: Baseline LFTs, CK only if symptoms; lipid panel to assess response (6–12 weeks after start).
- Counselling: Report muscle pain/weakness, dark urine; avoid grapefruit with atorvastatin. ([MDPI][10])
3) ACE inhibitor — Ramipril (example)
- Indication: Hypertension, albuminuric nephropathy, ASCVD risk reduction in some contexts. First-line for BP control in metabolic syndrome with diabetes/proteinuria.
- Mechanism: Inhibits angiotensin-converting enzyme → ↓angiotensin II → vasodilation, ↓aldosterone.
- Usual adult dosing: 2.5–10 mg once daily (typical start 2.5 mg daily, titrate).
- PK: Oral; metabolized to active metabolite; excreted renally.
- Adverse effects: Cough (class effect), hyperkalaemia, angioedema (rare), renal function decline in bilateral renal artery stenosis.
- Contraindications: Pregnancy, history of angioedema with ACEi, bilateral renal artery stenosis.
- Interactions: Potassium-sparing diuretics, potassium supplements → hyperkalaemia risk; NSAIDs may blunt antihypertensive effect and worsen renal function.
- Monitoring: Baseline and periodic creatinine/eGFR, potassium, BP.
- Counselling: Avoid pregnancy; report cough, facial swelling, lightheadedness. ([Ahad Journals][7])
4) Hydrochlorothiazide (HCTZ)
- Indication: Hypertension; commonly used in combination therapy.
- Mechanism: Thiazide diuretic — reduces sodium reabsorption in distal tubule → lowers blood pressure.
- Usual adult dosing: 12.5–50 mg once daily (start 12.5–25 mg).
- PK: Oral; renal excretion.
- Adverse effects: Hypokalaemia, hyponatraemia, hyperuricaemia (gout), hyperglycaemia (small effect), elevated cholesterol/TG (minor).
- Contraindications: Severe renal impairment for HCTZ; caution with gout.
- Interactions: Lithium (increases lithium levels), NSAIDs (reduce effect), other antihypertensives.
- Monitoring: Electrolytes, renal function, uric acid, BP.
- Counselling: Monitor for muscle cramps, palpitations (low K+), polyuria. ([Ahad Journals][7])
5) Fenofibrate
- Indication: Severe hypertriglyceridaemia (TG lowering), selected dyslipidaemia adjunct to statin when TG elevated.
- Mechanism: PPAR-α agonist → increases lipoprotein lipase activity → reduces TG.
- Usual adult dosing: 145 mg once daily (varies by formulation).
- PK: Oral; hepatic metabolism; renal excretion — dose adjust in renal impairment.
- Adverse effects: GI upset, elevated transaminases, risk of myopathy especially with statin (less than gemfibrozil).
- Contraindications: Severe hepatic impairment, severe renal impairment (adjust dose), gallbladder disease.
- Monitoring: LFTs, renal function, lipid panel, CK if muscle symptoms.
- Counselling: Report muscle pain/weakness, monitor for gallbladder symptoms. ([MDPI][10])
6) GLP-1 receptor agonist — Semaglutide (example)
- Indication: T2DM glycaemic control; obesity treatment (higher dose formulations). Beneficial weight loss and some CV outcome data (agent-specific).
- Mechanism: GLP-1 receptor agonist — increases glucose-dependent insulin secretion, slows gastric emptying, reduces appetite.
- Usual adult dosing: Varies by formulation and indication; subcutaneous weekly semaglutide for T2DM typically 0.25 → 0.5 → 1.0 mg weekly (for obesity, higher doses under specialist guidance). Follow product label.
- PK: Injectable; long half-life allows weekly dosing.
- Adverse effects: Nausea, vomiting, diarrhea, rarely pancreatitis; potential gallbladder disease risk; warning about medullary thyroid carcinoma (rodent data) — contraindicated in personal/family history of MEN2 or MTC for some GLP-1 RAs depending on label.
- Interactions: Slows gastric emptying — affects oral drug absorption (e.g., oral contraceptives, antibiotics) — counsel on consistent timing.
- Monitoring: GI tolerance, glucose, weight, signs of pancreatitis.
- Counselling: Expect transient GI side effects that often improve; report severe abdominal pain or persistent vomiting. Specialist initiation and monitoring usually recommended for obesity indications. ([MDPI][10])
7) Aspirin (low-dose)
- Indication: Secondary prevention of ASCVD. Primary prevention only in select high-risk patients after individualized risk/bleeding assessment.
- Usual adult dosing: 75–100 mg daily (varies by region/guideline).
- Important cautions: Bleeding risk; use individualized assessment.
- Monitoring / counselling: Watch for GI bleeding, bruising. ([Ahad Journals][7])
14) Special situations
- Pregnancy: metabolic syndrome components require specialist obstetric/endocrine care; many drugs (statins, ACEi) contraindicated. Focus on lifestyle and pregnancy-safe agents.
- Elderly / CKD: adjust drug choices/doses; avoid drugs that worsen renal function; involve specialists.
- Resource-limited settings: prioritize low-cost lifestyle programs, generic metformin, generic statin where indicated.
15) Practical patient-facing advice (brief)
- Losing 5–10% of body weight and exercising regularly often produces meaningful improvements in blood sugar, BP and lipids.
- Follow a Mediterranean-style diet, avoid sugary drinks and refined carbs, and consider structured programs (supervised diet/behavioural support).
- Medication may be needed to treat high BP, cholesterol, or blood sugar — drugs are chosen to lower cardiovascular risk, not just numbers.
- Screen family members and have regular checkups. ([ScienceDirect][8])
16) What’s new / evolving (brief)
- Interest in time-restricted eating and comprehensive habit-based lifestyle programs has grown; recent randomized trials (2023–2025) show promising metabolic benefits when combined with standard care. Newer diabetes/weight-loss agents (GLP-1 RAs) are increasingly used for obesity and to improve multiple metabolic syndrome components — cost and access remain issues. ([EatingWell][9])