Second Heart Sound (S2) — Detailed Explanation
1. What is S2?
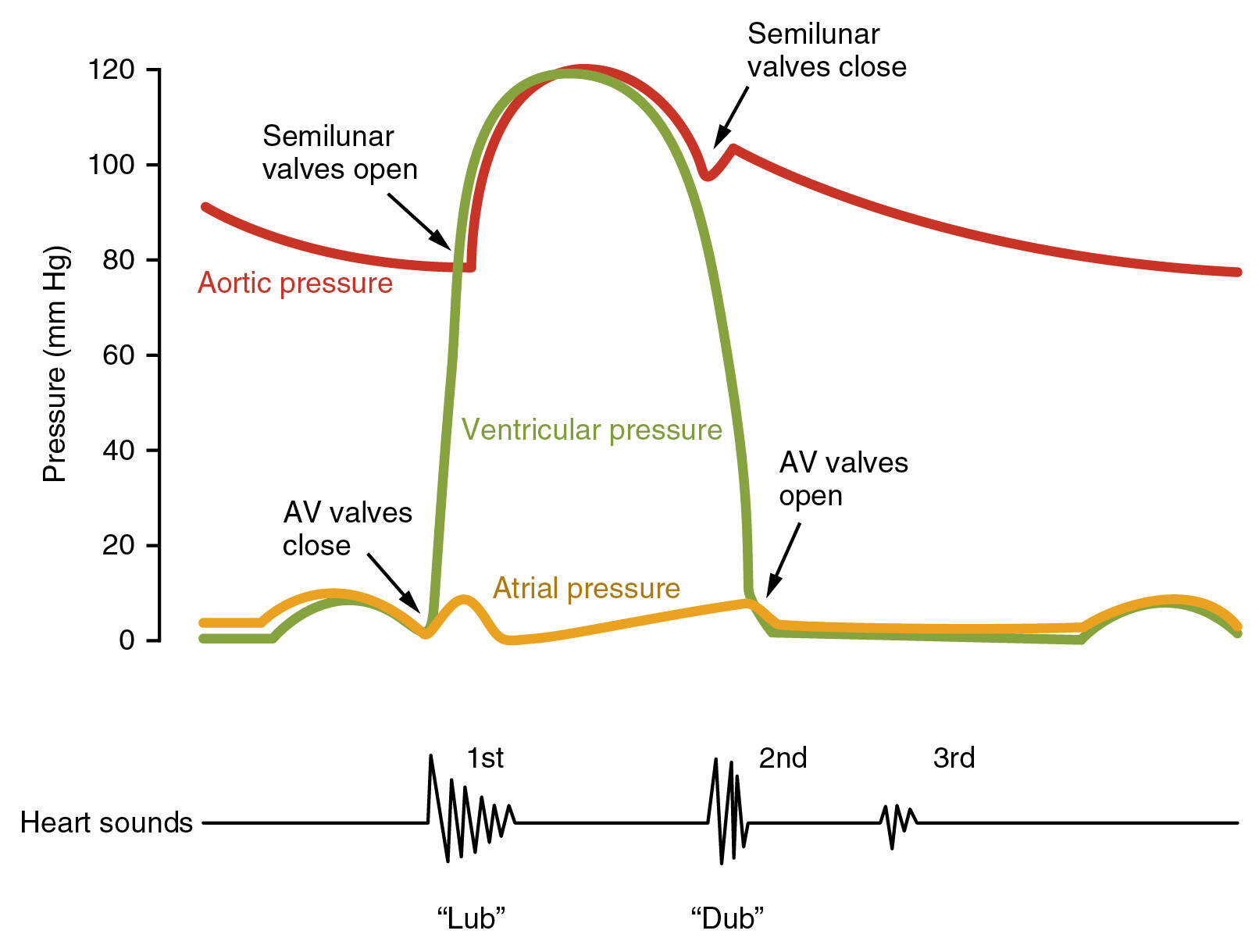
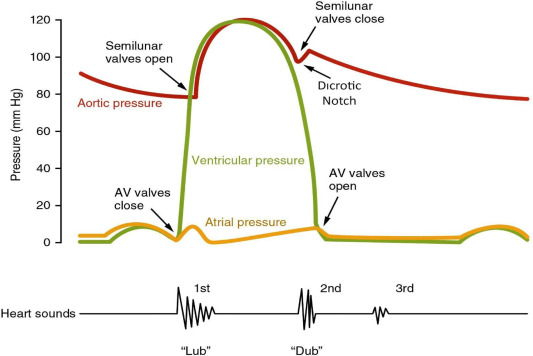
The second heart sound (S2) — heard as the “dub” in the classic “lub-dub” of a heartbeat — is produced by the closure of the semilunar valves at the end of ventricular systole and the beginning of ventricular diastole. These valves are:
- Aortic valve and
- Pulmonary valve. ([Wikipedia][1])
👉 External Link (Normal S2 overview):
• Heart sounds — Wikipedia → https://en.wikipedia.org/wiki/Heart_sounds ([Wikipedia][1])
2. Physiology — How S2 Is Produced
Heart sounds are vibrations created by blood flow and subsequent sudden valve closures. After the ventricles eject blood:
- The aortic valve closes when left ventricular pressure falls below aortic pressure,
- The pulmonary valve closes when right ventricular pressure falls below pulmonary artery pressure. These closures generate the S2 sound. ([NCBI][2])
S2 marks the end of mechanical systole and signals the beginning of diastole (ventricular relaxation). ([NCBI][3])
👉 External Link (Mechanism & details):
• The Second Heart Sound — Clinical Methods (NCBI) → https://www.ncbi.nlm.nih.gov/books/NBK341/ ([NCBI][3])
3. Components of S2
S2 consists of two components:
- Aortic component (A2) — closure of the aortic valve, and
- Pulmonic component (P2) — closure of the pulmonary valve. ([www.slideshare.net][4])
In healthy adults:
- A2 occurs slightly before P2.
- During expiration, S2 is usually heard as a single sound because A2 and P2 are very close. ([Stanford Medicine][5])
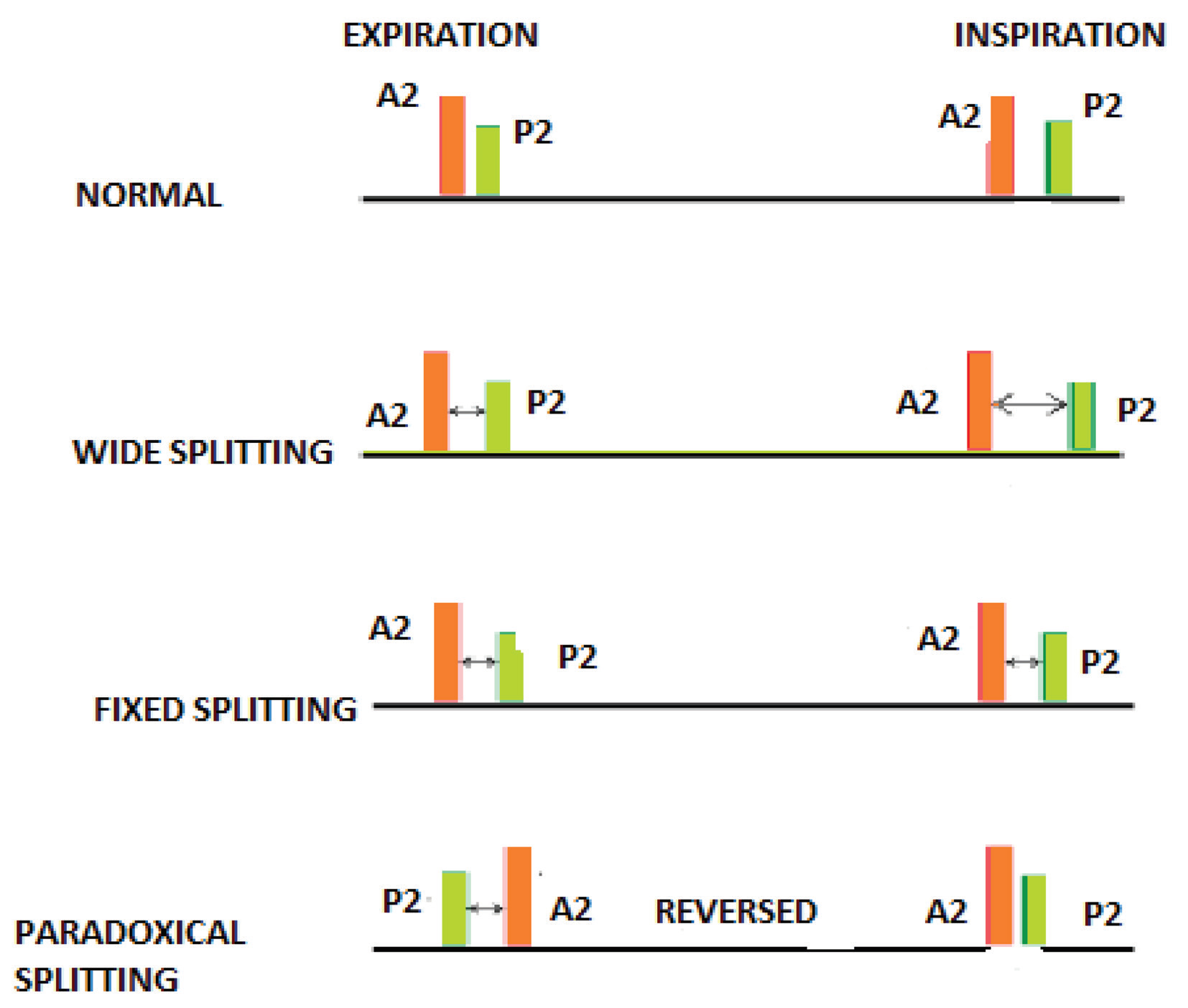
4. Splitting of Second Heart Sound
Splitting refers to hearing A2 and P2 separately instead of as a single sound.
Physiological (Normal) Splitting
During inspiration:
- Increased venous return to the right heart delays closure of the pulmonary valve (P2).
- The aortic valve (A2) closes first, so you hear two separate sounds (A2 then P2). ([Wikipedia][6])
This is normal splitting, best heard in the left upper sternal border. ([Wikipedia][6])
👉 External Link (Splitting in detail):
• Split S2 — Wikipedia → https://en.wikipedia.org/wiki/Split_S2 ([Wikipedia][6])
5. Clinical Variations of S2 Splitting
Changes in the pattern of S2 splitting can indicate pathology:
- Wide splitting: Seen in right bundle branch block (RBBB) or pulmonary stenosis — P2 is delayed. ([Wikipedia][6])
- Fixed splitting: Does not vary with respiration — characteristic of atrial septal defect (ASD). ([MSD Manuals][7])
- Paradoxical (reversed) splitting: P2 occurs before A2 — seen in left bundle branch block (LBBB) or aortic stenosis. ([Wikipedia][6])
👉 External Link (Pathological splitting details):
• Split S2 Heart Sound (MedZcool YouTube overview) → https://www.youtube.com/watch?v=98HM1fr3cq4 ([YouTube][8])
6. Auscultation — Where and How to Hear S2
- Use the diaphragm of the stethoscope. ([UW Departments][9])
- Best heard at the second right intercostal space for A2 and the second left intercostal space for P2. ([NCBI][3])
- S2 is typically high-pitched and shorter in duration than S1. ([NCBI][3])
👉 External Link (Auscultation technique):
• The Second Heart Sound (ResearchGate PDF) → https://www.researchgate.net/publication/49769873_The_Second_Heart_Sound ([ResearchGate][10])
7. Clinical Importance
S2 provides essential information about:
✔ Valvular function (especially semilunar valves),
✔ Conduction abnormalities,
✔ Right and left ventricular ejection timing,
✔ Intrathoracic pressure effects (e.g., with respiration). ([IJCDW][11])
Abnormalities in the intensity, timing, or pattern of S2 may point toward cardiac diseases that need further evaluation (e.g., echocardiography). ([Wikipedia][1])
Summary
| Feature | Description |
| ---------------------- | ----------------------------------------------- |
| Sound | “Dub” of “lub-dub” |
| Cause | Closure of semilunar valves (aortic & pulmonic) |
| Components | A2 (aortic), P2 (pulmonic) |
| Normal Splitting | More noticeable on inspiration |
| Clinical Variation | Wide, fixed, paradoxical splits |
| Best Heard | 2nd intercostal spaces |
If you want, I can also provide audio examples of S2 and its pathological variants with descriptions.
[1]: https://en.wikipedia.org/wiki/Heart_sounds?utm_source=chatgpt.com "Heart sounds"
[2]: https://www.ncbi.nlm.nih.gov/books/NBK541010/?utm_source=chatgpt.com "Physiology, Heart Sounds - StatPearls"
[3]: https://www.ncbi.nlm.nih.gov/books/NBK341/?utm_source=chatgpt.com "The Second Heart Sound - Clinical Methods"
[4]: https://www.slideshare.net/slideshow/fffffs2/33993054?utm_source=chatgpt.com "second heart sound | PPT"
[5]: https://med.stanford.edu/stanfordmedicine25/the25/cardiac.html?utm_source=chatgpt.com "Cardiac Second Heart Sounds - Stanford Medicine"
[6]: https://en.wikipedia.org/wiki/Split_S2?utm_source=chatgpt.com "Split S2"
[7]: https://www.msdmanuals.com/professional/multimedia/audio/split_2nd_heart_sound_atrial_septal_defect?utm_source=chatgpt.com "Split Second Heart Sound With Atrial Septal Defect"
[8]: https://www.youtube.com/watch?v=98HM1fr3cq4&utm_source=chatgpt.com "Fixed Split S2 - Heart Sounds - MEDZCOOL - YouTube"
[9]: https://depts.washington.edu/physdx/heart/tech1.html?utm_source=chatgpt.com "Techniques - Heart Sounds & Murmurs Exam"
[10]: https://www.researchgate.net/publication/49769873_The_Second_Heart_Sound?utm_source=chatgpt.com "(PDF) The Second Heart Sound"
[11]: https://ijcdw.org/second-heart-sound/?utm_source=chatgpt.com "Second Heart Sound"
Below is an expanded, exam-oriented set of 25 clinical case scenarios on the Second Heart Sound (S2) that explicitly includes the conditions you asked for: LVF, ASD, VSD, WPW, Coxsackie B myocarditis, chronic anemia, wet beriberi, and other important causes.
Each scenario highlights how S2 is affected and why.
Second Heart Sound (S2): 25 Comprehensive Case Scenarios
1. Normal Physiological Splitting
A 20-year-old healthy male has S2 splitting on inspiration that disappears on expiration.
Mechanism: Delayed P2 due to increased venous return.
2. Fixed Split S2 – ASD
A 26-year-old woman has wide S2 splitting that does not vary with respiration.
Diagnosis: Atrial septal defect (ASD).
3. ASD with Pulmonary Hypertension
A 40-year-old untreated ASD patient has a loud, single S2.
Cause: Pulmonary hypertension causing loud P2 merging with A2.
4. Wide Split S2 – VSD with Increased Flow
A child with pansystolic murmur and heart failure signs has wide S2 splitting.
Mechanism: Increased RV stroke volume delaying P2.
5. Single Loud S2 – Eisenmenger Syndrome
A cyanotic adult with long-standing VSD has a single loud S2.
Reason: Severe pulmonary hypertension abolishing split.
6. Paradoxical Splitting – Left Ventricular Failure (LVF)
A patient with dilated cardiomyopathy has S2 split during expiration.
Cause: Prolonged LV ejection delaying A2.
7. Soft S2 in Severe LVF
A patient in cardiogenic shock has faint S2.
Reason: Reduced pressure gradients across semilunar valves.
8. Loud P2 in LVF with Pulmonary Hypertension
A chronic LVF patient develops loud P2.
Cause: Secondary pulmonary hypertension.
9. Paradoxical Splitting – WPW Syndrome
A young adult with delta waves on ECG has reversed S2 splitting.
Mechanism: Pre-excitation altering ventricular activation and delaying A2.
10. Wide Split S2 – Right Bundle Branch Block
A patient with syncope has wide inspiratory splitting of S2.
Cause: Delayed RV depolarization → delayed P2.
11. Coxsackie B Myocarditis
A young adult post-viral illness has soft S2.
Mechanism: Depressed myocardial contractility reduces valve closure intensity.
12. Acute Myocarditis with LV Dysfunction
A febrile patient develops heart failure and paradoxical S2 splitting.
Cause: Delayed LV systole affecting A2.
13. Chronic Anemia
A patient with Hb 6 g/dL has loud S2.
Reason: Hyperdynamic circulation with forceful valve closure.
14. Wet Beriberi (High-Output Failure)
A malnourished patient with edema and tachycardia has loud S2.
Mechanism: High-output cardiac state increases valve closure velocity.
15. Pulmonary Hypertension
A patient with progressive dyspnea has loud P2.
Key Sign: Accentuated pulmonic component of S2.
16. Severe Pulmonary Hypertension – Single S2
A patient with idiopathic PAH has single palpable S2.
Explanation: Very loud P2 masks A2.
17. Aortic Stenosis
An elderly patient with syncope has soft or absent A2.
Cause: Calcified, immobile aortic valve.
18. Severe Aortic Stenosis – Single S2
A patient with severe AS has single S2.
Reason: Absent A2.
19. Aortic Regurgitation
A patient with bounding pulse has soft A2.
Mechanism: Incomplete valve closure.
20. Mitral Stenosis
A rheumatic patient has loud P2.
Cause: Pulmonary hypertension secondary to MS.
21. Pulmonary Stenosis
A young adult with ejection systolic murmur has wide S2 split.
Mechanism: Delayed P2.
22. COPD
A smoker with hyperinflated chest has soft P2.
Reason: Poor sound transmission through lungs.
23. Acute Pulmonary Embolism
A patient with sudden dyspnea and chest pain has loud P2.
Cause: Acute rise in pulmonary artery pressure.
24. Cardiogenic Shock
A post-MI patient in shock has barely audible S2.
Reason: Severely reduced cardiac output.
25. Hyperthyroidism / High-Output State
A thyrotoxic patient has loud S2.
Mechanism: Increased flow velocity and contractility.
Conditions Explicitly Covered
✔ LVF
✔ ASD
✔ VSD
✔ WPW
✔ Coxsackie B myocarditis
✔ Chronic anemia
✔ Wet beriberi
✔ Pulmonary hypertension
✔ Valve stenosis & regurgitation
✔ Conduction disorders
✔ High-output & low-output states