1. Definition
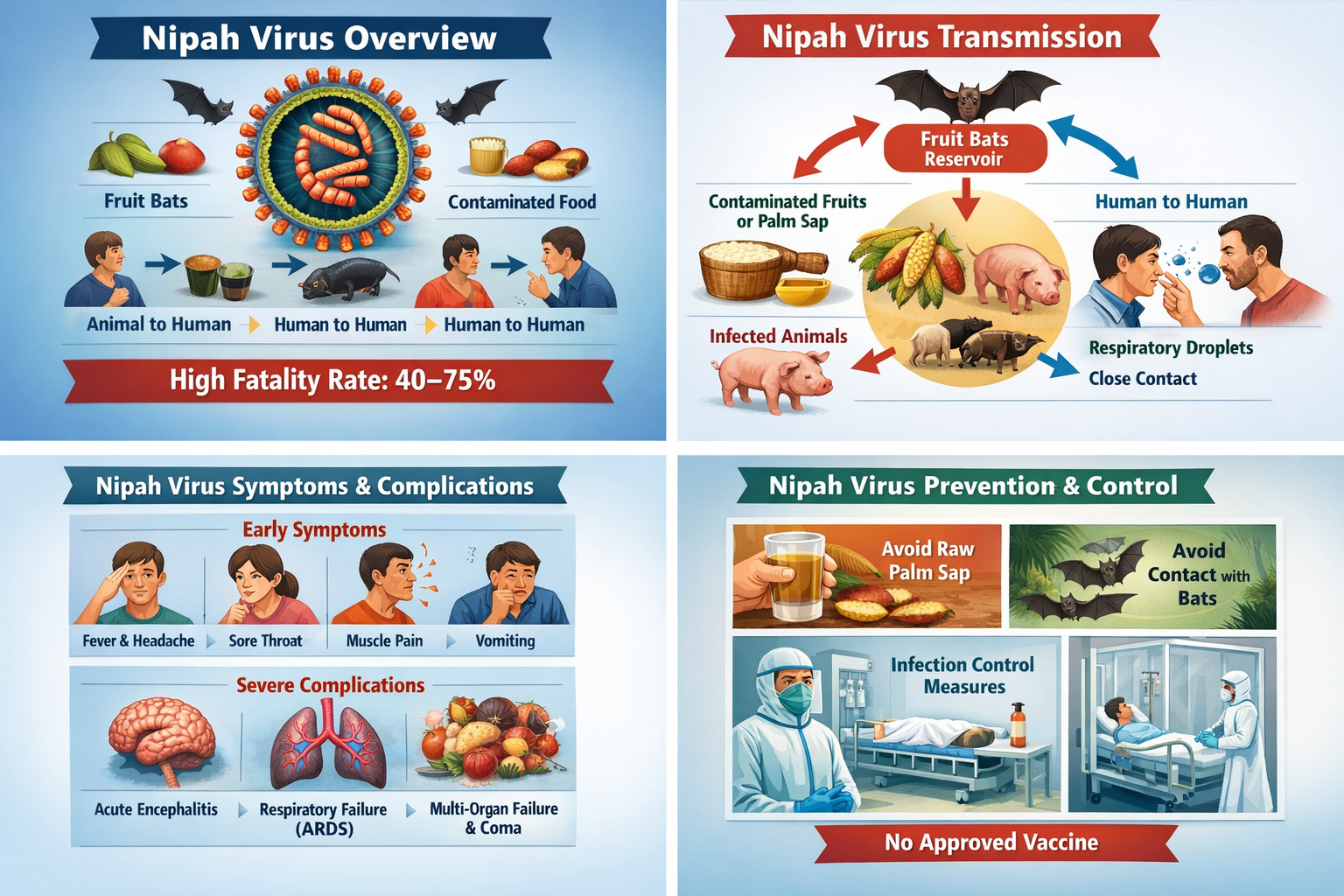
Nipah virus (NiV) is a highly pathogenic zoonotic virus belonging to the Paramyxoviridae family that causes severe encephalitis and acute respiratory illness in humans, with a high case fatality rate (40–75%).
2. Virology & Pathophysiology
- Virus type: Negative-sense, single-stranded RNA virus
- Natural reservoir: Fruit bats (Pteropus species)
- Mechanism of disease:
* Virus enters through respiratory or oral mucosa
* Spreads via bloodstream
* Infects endothelial cells, causing vasculitis
* Leads to brain inflammation (encephalitis) and respiratory damage
* Causes multi-organ failure in severe cases
3. Transmission Routes
- Bat → Human (via contaminated fruits or palm sap)
- Animal (pigs) → Human
- Human → Human (respiratory droplets & close contact)
- Exposure to body fluids of infected individuals
4. Incubation Period
4–14 days (can extend up to 45 days)
5. Clinical Features
A. Early Symptoms
- Fever
- Headache
- Myalgia
- Fatigue
- Sore throat
- Vomiting
B. Neurological Manifestations
- Confusion
- Altered mental status
- Seizures
- Reduced consciousness
- Encephalitis
- Coma
C. Respiratory Symptoms
- Cough
- Shortness of breath
- Acute respiratory distress syndrome (ARDS)
D. Severe Complications
- Brain edema
- Multi-organ failure
- Shock
- Death
6. Long-Term Sequelae (Survivors)
- Memory impairment
- Personality changes
- Chronic fatigue
- Seizure disorders
- Relapsing encephalitis months to years later
7. Risk Factors
- Consumption of raw date palm sap
- Contact with bats or infected animals
- Healthcare workers without PPE
- Close contact with infected patients
- Living in outbreak regions (South Asia)
8. Diagnosis & Investigations
A. Laboratory Confirmation
- RT-PCR (Gold Standard) — throat swab, CSF, blood
- ELISA — IgM and IgG antibodies
- Virus isolation (BSL-4 labs only)
B. Supportive Tests
- CBC (may show thrombocytopenia)
- Liver function tests
- CSF analysis
- MRI brain → encephalitis lesions
- Chest X-ray/CT → pneumonia or ARDS
9. Differential Diagnoses
- Japanese encephalitis
- COVID-19
- Dengue encephalopathy
- Herpes simplex encephalitis
- West Nile virus
- Bacterial meningitis
- Rabies
10. Management & Treatment
No specific approved antiviral therapy. Management is supportive.
A. Supportive Care
- ICU monitoring
- Oxygen therapy
- Mechanical ventilation if respiratory failure
- IV fluids and electrolyte balance
- Antipyretics and analgesics
- Seizure control
- Nutritional support
11. Antiviral & Experimental Therapies
Ribavirin
- Indication: Experimental use
- Mechanism: RNA synthesis inhibition
- Dose (Adult): 30 mg/kg loading → 15 mg/kg every 6 hours
- Adverse effects: Hemolytic anemia, liver toxicity
Monoclonal Antibody (m102.4)
- Experimental neutralizing antibody
- Used under compassionate protocols
Remdesivir
- Investigational use in some outbreaks
12. Infection Control & Isolation
- Strict contact and droplet precautions
- PPE for healthcare workers
- Patient isolation
- Safe disposal of body fluids
- Avoid unnecessary exposure
13. Prevention Measures
Public Health Measures
- Avoid raw date palm sap
- Wash fruits thoroughly
- Avoid sick animals
- Community awareness
Hospital Measures
- Early detection
- Quarantine contacts
- Strict hygiene protocols
Vaccine Status
- No approved vaccine yet
- Several vaccines in clinical trials
14. Mortality & Prognosis
- Fatality Rate: 40–75%
- Worse outcomes with:
* Rapid neurological deterioration
* Respiratory failure
* Delayed hospitalization
15. Global & Indian Context
- First outbreak: Malaysia (1998)
- Recurrent outbreaks: Bangladesh, India (Kerala)
- WHO classifies Nipah virus as a priority pandemic threat
16. Patient Counselling Points
- Seek urgent care for fever + confusion
- Avoid contact with suspected cases
- Follow hygiene & infection prevention
- Survivors may need long-term neurological follow-up
17. Key Takeaway Summary
- Nipah virus is a highly fatal zoonotic encephalitis virus
- Spreads via bats, animals, and human contact
- Causes brain inflammation & respiratory failure
- No cure yet — prevention & early supportive care save lives