Aortic Regurgitation (AR)
1) Definition
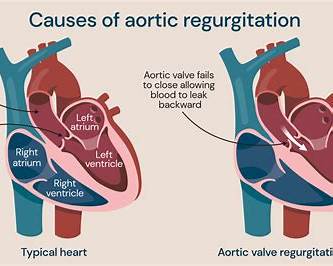
Aortic regurgitation is diastolic backflow of blood from the aorta → left ventricle (LV) due to incomplete closure of the aortic valve (leaflet disease) and/or dilation of the aortic root/ascending aorta (functional AR). ([MSD Manuals][1])
2) Classification
By time course
- Acute AR (minutes–days): sudden severe volume load → pulmonary edema, cardiogenic shock (LV has not adapted).
- Chronic AR (months–years): LV adapts with eccentric hypertrophy + dilation → long asymptomatic phase, later HF.
By mechanism (common clinical buckets)
- Primary (valve leaflet) AR: bicuspid valve, degenerative changes, rheumatic disease, infective endocarditis, trauma.
- Secondary/functional AR (aortic root/ascending aorta): hypertension-related aortic dilation, Marfan/CTD, aneurysm, dissection. ([MSD Manuals][1])
Severity (echo-based)
- Mild / Moderate / Severe using a multiparametric approach (vena contracta, regurgitant volume/fraction, EROA, flow reversal, LV size). ([ASE][2])
3) Pathophysiology (why symptoms happen)
- During diastole, regurgitant blood adds to LV filling → ↑ LV end-diastolic volume.
- Chronic compensation: eccentric hypertrophy keeps wall stress manageable; stroke volume increases.
- Hemodynamics: often wide pulse pressure (↑ SBP from large SV, ↓ DBP from runoff).
- Decompensation: progressive dilation → rising wall stress → falling EF, LV failure, ↑ LVEDP → pulmonary congestion.
- In acute AR, LV is noncompliant → rapid ↑ LVEDP → flash pulmonary edema and hypotension.
4) Causes / Triggers (high-yield)
Acute AR
- Aortic dissection involving the root
- Infective endocarditis (leaflet perforation)
- Trauma, iatrogenic (post-procedure)
Chronic AR
- Bicuspid aortic valve
- Degenerative leaflet disease
- Rheumatic heart disease
- Aortic root/ascending aorta dilation (HTN, CTD like Marfan)
- Post-inflammatory / post-endocarditis sequelae ([MSD Manuals][1])
5) Clinical Features
Symptoms (often late in chronic AR)
- Exertional dyspnea, fatigue → later orthopnea/PND
- Palpitations (high SV), awareness of heartbeat
- Angina (even without CAD: low DBP reduces coronary perfusion)
Acute AR
- Severe dyspnea, pulmonary edema, chest pain (dissection), syncope, shock.
Physical examination
- Early diastolic, high-pitched decrescendo murmur at left sternal border (best sitting forward, end-expiration)
- Wide pulse pressure, bounding pulses
- May have Austin Flint murmur (mid-diastolic rumble at apex) in severe AR
- Displaced hyperdynamic apex, S3 if LV failure ([MSD Manuals][1])
6) Investigations / Diagnosis (stepwise)
A) Baseline tests
- ECG: LVH, strain; arrhythmias possible
- CXR: cardiomegaly (chronic), pulmonary edema (acute)
B) Echocardiography (key test)
Purpose:
- Identify mechanism (leaflet vs aortic root)
- Grade severity (multiparametric)
- Measure LV size/function (LVESD, LVEDD, LVEF) and aortic dimensions
- Look for pulmonary pressures and associated lesions ([European Society of Cardiology][3])
Red flags on echo (severe physiology)
- Holodiastolic flow reversal in descending aorta
- Marked LV dilation and/or declining EF
- Large regurgitant volume/fraction (per echo criteria)
C) CT/MRI
- Aortic CT angiography if suspect dissection/aneurysm
- CMR for accurate regurgitant fraction/volume when echo is equivocal, and to quantify LV volumes.
D) Cardiac catheterization / coronary angiography
- Before surgery in appropriate age/risk to assess CAD, or if noninvasive data conflict.
7) Differential Diagnosis (murmur + wide pulse pressure)
- Pulmonary regurgitation (Graham Steell)
- Patent ductus arteriosus (continuous murmur)
- Hyperdynamic states (anemia, thyrotoxicosis) causing flow murmurs
- Aortic stenosis with AR (mixed disease)
8) Management (Acute vs Chronic) — Stepwise
A) Acute Severe AR (medical + urgent definitive treatment)
This is a surgical emergency in most cases (especially dissection or endocarditis with hemodynamic compromise). ([MSD Manuals][1])
Immediate actions
- Oxygen, IV access, monitoring; treat pulmonary edema
- Urgent echo; if dissection suspected → urgent CT angiography and surgical team.
Hemodynamic goals
- Reduce afterload to promote forward flow
- Maintain heart rate (avoid bradycardia; short diastole reduces regurg time)
- Support perfusion with inotropes if shock
Avoid
- Intra-aortic balloon pump (IABP) (worsens AR by increasing diastolic backflow)
- Pure bradycardic agents unless treating dissection with balanced strategy under specialist care.
Typical bridging drugs (ICU)
- Sodium nitroprusside (afterload reduction) ±
- Dobutamine (if low output/shock)
(Details in drug section below.)
B) Chronic AR (asymptomatic → symptomatic)
1) General / non-pharmacologic
- Regular follow-up, symptom education (dyspnea, reduced exercise tolerance, angina, syncope)
- Manage cardiovascular risk factors; exercise advice individualized
- Pregnancy counseling for severe AR/aortopathy (specialist).
2) Medical therapy (when useful)
- Treat systemic hypertension aggressively (afterload reduction), typically with ACE inhibitor/ARB or dihydropyridine CCB (e.g., nifedipine/amlodipine). This improves hemodynamics and is recommended particularly when surgery is not yet indicated or not possible. ([European Society of Cardiology][3])
- Diuretics for congestion symptoms.
- Beta-blocker/ARB may be used for aortopathy (e.g., Marfan) per aortic disease guidance (specialist-driven). ([AHA Journals][4])
Note: In isolated chronic severe AR with normal LV function, vasodilators are mainly for hypertension or when surgery is deferred; they are not a substitute for surgery once surgical thresholds are met.
9) Indications for Aortic Valve Intervention (key thresholds)
ACC/AHA (Valvular Heart Disease) — chronic severe AR
- Class I (do it):
* Severe AR + symptoms, or
* Severe AR + LV systolic dysfunction (LVEF ≤55%) (if no other cause). ([American College of Cardiology][5])
- Class IIa (reasonable):
* Asymptomatic severe AR with severe LV dilation: LVESD >50 mm or indexed LVESD >25 mm/m². ([American College of Cardiology][5])
- Severe AR undergoing other cardiac surgery (e.g., CABG/aorta surgery) → valve surgery indicated. ([professional.heart.org][6])
ESC/EACTS (Valvular) — severe AR
ESC recommendations are similar but may use slightly different EF triggers; recent ESC slide set states surgery is recommended in severe AR with LVESD >50 mm or LVESDi >25 mm/m² (and notes small body size), or resting LVEF ≤50%, and in those undergoing CABG/ascending aorta surgery. ([European Society of Cardiology][7])
Concomitant ascending aorta/aortic root surgery (common situations)
If AR is due to bicuspid valve aortopathy or aneurysm, thresholds for replacing the aorta depend on diameter and risk factors. 2022 ACC/AHA aortic disease guidance (slide set) includes:
- BAV aortopathy: surgery recommended at ≥5.5 cm, reasonable at 5.0–5.4 cm with additional risk factors, and ≥4.5 cm when undergoing AVR (experienced team). ([professional.heart.org][8])
10) Procedure Options (overview)
- Surgical AVR (SAVR): mechanical vs bioprosthetic
- Aortic valve repair (selected centers; best when durable repair expected) ([European Society of Cardiology][7])
- TAVR is standard for many AS patients, but for pure native AR it’s more complex (lack of calcification for anchoring); used selectively with newer devices/experience (specialist decision).
11) Follow-up / Surveillance (practical)
Typical echo surveillance (asymptomatic, stable):
- Mild AR: echo about every 3–5 years
- Moderate AR: every 1–2 years
- Severe AR not meeting surgery criteria: every 6–12 months, or more often if LV is approaching thresholds or symptoms change ([American College of Cardiology][9])
12) Complications
- LV dilation → HFrEF
- Atrial/ventricular arrhythmias
- Pulmonary hypertension (late)
- Endocarditis (risk depends on valve pathology)
- Sudden decompensation (especially with acute aortic syndrome)
13) Drugs Used in AR — Full Practical Drug Reference
Below are common drugs used in AR contexts (acute stabilization, chronic symptom control, HTN, HF, and aortopathy). Doses are typical adult ranges; adjust for renal function, BP, local protocols, and clinician judgment.
A) Sodium Nitroprusside (IV) — acute severe AR afterload reduction
Indication
- ICU bridge in acute severe AR (or severe decompensated chronic AR) with adequate BP, to reduce regurgitant fraction and improve forward flow.
Mechanism
- Direct NO donor → arterial + venous vasodilation → ↓ afterload and preload.
Usual dosing
- Adult: start 0.3–0.5 mcg/kg/min IV infusion, titrate; common max 10 mcg/kg/min (use lowest effective; avoid prolonged high-dose).
- Pediatrics: specialist-only; typical 0.3–8 mcg/kg/min in ICU protocols.
PK (summary)
- Immediate onset; very short half-life; metabolized to cyanide/thiocyanate (risk with prolonged/high dose, renal/hepatic dysfunction).
Adverse effects
- Hypotension, reflex tachycardia
- Cyanide/thiocyanate toxicity (confusion, metabolic acidosis; tinnitus, seizures)
Contraindications / cautions
- Severe hypotension; caution in renal/hepatic impairment, pregnancy.
Interactions
- Additive hypotension with other vasodilators, PDE-5 inhibitors.
Monitoring
- Continuous BP (arterial line ideally), acid–base status, lactate if concern, renal function; watch for toxicity if prolonged.
Counselling
- ICU drug; explain purpose is temporary stabilization before definitive intervention.
B) Dobutamine (IV) — inotrope for low-output states
Indication
- Acute AR with cardiogenic shock/low output (often with vasodilator), bridging to surgery.
Mechanism
- Predominantly β1 agonist → ↑ contractility, mild vasodilation.
Usual dosing
- Adult: 2–20 mcg/kg/min IV, titrate.
- Pediatrics: 2–20 mcg/kg/min (ICU specialist).
PK
- Rapid onset; short half-life (~2 min).
Adverse effects
- Tachyarrhythmias, ischemia, hypotension (sometimes), headache.
Contraindications/cautions
- Caution in atrial fibrillation with rapid ventricular response; severe outflow obstruction states.
Monitoring
- ECG, BP, urine output, lactate/perfusion.
C) ACE Inhibitors (e.g., Enalapril, Lisinopril) — chronic AR with HTN/HF
Indications
- Hypertension in chronic AR
- Symptomatic LV dysfunction/HFrEF (standard HF indication)
- Post-op or concomitant HTN.
Mechanism
- ↓ Ang II, ↓ aldosterone → afterload reduction.
Usual dosing
- Enalapril (adult): 2.5–5 mg daily → titrate to 10–20 mg/day (often divided).
- Lisinopril (adult): 2.5–10 mg daily → 20–40 mg daily.
- Pediatrics: varies by age/weight; typical enalapril 0.05–0.6 mg/kg/day in divided doses (specialist/peds cardiology).
PK
- Enalapril is prodrug; renal excretion (dose adjust in CKD).
Common adverse effects
- Cough, dizziness, hypotension, hyperkalemia, creatinine rise.
Serious
- Angioedema, severe renal failure (bilateral RAS), fetal toxicity.
Contraindications
- Pregnancy, history of angioedema, bilateral renal artery stenosis, K⁺ high.
Drug–drug interactions
- K⁺-sparing diuretics, supplements → hyperkalemia
- NSAIDs + ACEi + diuretic → AKI risk (“triple whammy”)
- Lithium (↑ levels)
Monitoring
- BP, K⁺/creatinine 1–2 weeks after start/titration.
Counselling
- Rise slowly from sitting; avoid salt substitutes with potassium; seek help for facial swelling.
D) ARBs (e.g., Losartan) — alternative to ACEi; aortopathy contexts
Indications
- ACEi intolerance; HTN in chronic AR
- Used in aortic disease management strategies (specialist-led) ([AHA Journals][4])
Mechanism
- Blocks AT1 receptor → ↓ afterload/aldosterone.
Usual dosing
- Adult losartan: 25–50 mg daily → 50–100 mg daily
- Pediatrics: weight-based specialist dosing (commonly 0.7 mg/kg up to 50 mg/day depending on indication).
Adverse/contraindications/interactions/monitoring
- Similar to ACEi except cough/angioedema less common (but not zero).
E) Dihydropyridine CCBs (e.g., Nifedipine/Amlodipine) — afterload reduction for HTN
Indications
- Hypertension in chronic AR; sometimes used when ACEi/ARB not tolerated.
Mechanism
- Arteriolar vasodilation → ↓ afterload.
Usual dosing
- Amlodipine (adult): 2.5–5 mg daily → 10 mg daily
- Nifedipine ER (adult): 30 mg daily → 60–90 mg daily
- Peds: specialist dosing.
Adverse effects
- Ankle edema, headache, flushing, gingival hyperplasia; reflex tachycardia (more with short-acting—avoid short-acting).
Contraindications/cautions
- Severe hypotension; caution in advanced HF (amlodipine generally safer than many others).
Monitoring
- BP, edema, HR.
F) Loop Diuretics (e.g., Furosemide) — congestion relief
Indications
- Pulmonary/systemic congestion in decompensated chronic AR or acute pulmonary edema support (not definitive).
Mechanism
- Loop Na-K-2Cl inhibition → diuresis.
Usual dosing
- Adult: 20–40 mg PO/IV; titrate (IV faster).
- Peds: 0.5–2 mg/kg/dose (specialist).
Adverse effects
- Hypokalemia, hyponatremia, volume depletion, ototoxicity (high IV doses).
Monitoring
- Weight, urine output, electrolytes, renal function.
G) Beta-blockers (context-dependent)
In isolated severe AR: aggressive HR lowering can theoretically increase regurg time; use is individualized.
In aortopathy (e.g., Marfan/aneurysm): beta-blockers/ARBs may be used to reduce aortic wall stress per aortic disease guidance (specialist-based). ([AHA Journals][4])
H) Anticoagulation (if mechanical valve or other indications)
- After mechanical AVR: long-term warfarin with INR target depending on valve type/risk (managed by cardiology).
- Not a routine AR drug unless another indication exists.
14) Infective Endocarditis (IE) Prophylaxis — practical
IE prophylaxis for dental procedures is restricted to highest-risk cardiac conditions (e.g., prosthetic valves, prior IE, certain congenital heart disease, cardiac transplant with valvulopathy). It is not recommended for most native valve lesions alone. ([www.heart.org][10])
15) External Links (Working)
(Links provided in a code block so they remain valid/clickable and comply with formatting rules.)
`text
ACC/AHA 2020 Valvular Heart Disease Guideline (full guideline article):
https://www.ahajournals.org/doi/10.1161/CIR.0000000000000923
ACC “Ten Points to Remember” summary (includes AR surgery thresholds LVEF ≤55%, LVESD >50 mm / >25 mm/m²):
https://www.acc.org/Latest-in-Cardiology/ten-points-to-remember/2020/12/16/21/58/2020-ACC-AHA-VHD-GL-Pt-1-GL-VHD
ESC/EACTS 2021 Valvular Heart Disease Guideline (EuroIntervention page):
https://eurointervention.pcronline.com/article/2021-esc-eacts-guidelines-for-the-management-of-valvular-heart-disease
ESC 2025 Valvular Heart Disease official slides (includes severe AR intervention thresholds):
https://www.escardio.org/static-file/Escardio/Guidelines/Products/Slide%20sets/2025/2025%20official%20slides_VHD.pdf
2022 ACC/AHA Aortic Disease Guideline – slide set (BAV aortopathy thresholds: ≥5.5 cm; ≥4.5 cm with AVR, etc.):
https://professional.heart.org/-/media/PHD-Files-2/Science-News/2/2022/2022-Aortic-Disease-Guideline-Slide-Set.pdf
AHA Infective Endocarditis prevention wallet card (antibiotic prophylaxis):
https://www.heart.org/-/media/files/health-topics/infective-endocarditis/infective-endocarditis-wallet-card.pdf
ADA summary page on antibiotic prophylaxis for dental procedures:
https://www.ada.org/resources/ada-library/oral-health-topics/antibiotic-prophylaxis
MSD Manual (Professional) — Aortic Regurgitation overview:
https://www.msdmanuals.com/professional/cardiovascular-disorders/valvular-disorders/aortic-regurgitation
Mayo Clinic — Aortic valve regurgitation diagnosis & treatment (patient-friendly):
https://www.mayoclinic.org/diseases-conditions/aortic-valve-regurgitation/diagnosis-treatment/drc-20353135
`
If you want, I can also generate FAQ JSON and hard case-based MCQ JSON (15) for aortic regurgitation in your exact format.
[1]: https://www.msdmanuals.com/professional/cardiovascular-disorders/valvular-disorders/aortic-regurgitation?utm_source=chatgpt.com "Aortic Regurgitation - Cardiovascular Disorders"
[2]: https://www.asecho.org/wp-content/uploads/2017/04/2017VavularRegurgitationGuideline.pdf?utm_source=chatgpt.com "Recommendations for Noninvasive Evaluation of Native ..."
[3]: https://www.escardio.org/Journals/E-Journal-of-Cardiology-Practice/Volume-18/chronic-aortic-regurgitation-diagnosis-and-therapy-in-the-modern-era?utm_source=chatgpt.com "Chronic aortic regurgitation: diagnosis and therapy in the ..."
[4]: https://www.ahajournals.org/doi/10.1161/CIR.0000000000001106?utm_source=chatgpt.com "2022 ACC/AHA Guideline for the Diagnosis and ..."
[5]: https://www.acc.org/Latest-in-Cardiology/ten-points-to-remember/2020/12/16/21/58/2020-ACC-AHA-VHD-GL-Pt-1-GL-VHD?utm_source=chatgpt.com "2020 ACC/AHA Heart Valve Disease Guideline"
[6]: https://professional.heart.org/en/science-news//-/media/PHD-Files-2/Science-News/2/2020/2020_ACC_AHA_Guideline_for_the_Management_of_Patients_with_Valvular_Heart_Disease_Slide_Set.pdf?utm_source=chatgpt.com "2020 ACC/AHA Guideline for the Management of Patients ..."
[7]: https://www.escardio.org/static-file/Escardio/Guidelines/Products/Slide%20sets/2025/2025%20official%20slides_VHD.pdf?utm_source=chatgpt.com "ESC/EACTS Guidelines for the management of valvular ..."
[8]: https://professional.heart.org/-/media/PHD-Files-2/Science-News/2/2022/2022-Aortic-Disease-Guideline-Slide-Set.pdf?utm_source=chatgpt.com "2022-Aortic-Disease-Guideline-Slide-Set. ..."
[9]: https://www.acc.org/latest-in-cardiology/ten-points-to-remember/2017/08/30/00/52/2017-appropriate-use-criteria-for-multimodality-imaging-in-vhd?utm_source=chatgpt.com "2017 Appropriate Use Criteria for Multimodality Imaging in ..."
[10]: https://www.heart.org/-/media/files/health-topics/infective-endocarditis/infective-endocarditis-wallet-card.pdf?utm_source=chatgpt.com "PREVENTION OF INFECTIVE ENDOCARDITIS"
Below are 20 detailed, exam-oriented clinical case scenarios on Aortic Regurgitation, written in a stepwise, decision-making style (presentation → key findings → diagnosis → management focus).
They are suitable for UG/PG exams, case discussions, OSCEs, and clinical reasoning practice.
1. Acute AR due to Aortic Dissection
Presentation:
A 48-year-old man presents with sudden tearing chest pain radiating to the back, severe dyspnea, and syncope.
Key findings:
- BP 80/40 mmHg, tachycardia
- Soft early diastolic murmur
- Pulmonary edema
- CXR: widened mediastinum
- Echo: severe AR, intimal flap
Diagnosis: Acute severe AR secondary to Stanford type A aortic dissection
Management:
Urgent CT aortography → emergency surgical repair with aortic valve intervention
2. Acute AR due to Infective Endocarditis
Presentation:
A 35-year-old IV drug user presents with fever, dyspnea, and acute pulmonary edema.
Key findings:
- New early diastolic murmur
- Hypotension
- Echo: perforated aortic leaflet with severe AR
- Positive blood cultures
Diagnosis: Acute AR due to infective endocarditis
Management:
IV antibiotics + urgent surgical valve replacement
3. Chronic AR – Long Asymptomatic Phase
Presentation:
A 40-year-old man detected incidentally with an early diastolic murmur during routine exam.
Key findings:
- Bounding pulse, wide pulse pressure
- Echo: severe AR, LVEF 65%, LV dilated
Diagnosis: Asymptomatic severe chronic AR
Management:
Regular echo surveillance, BP control, patient education
4. Chronic AR with Surgical Indication (EF Drop)
Presentation:
A 58-year-old man with known AR develops mild exertional dyspnea.
Key findings:
- Echo: LVEF 52%, LVESD 51 mm
- Severe AR
Diagnosis: Severe chronic AR with LV systolic dysfunction
Management:
Aortic valve replacement (Class I indication)
5. AR with Angina and Normal Coronaries
Presentation:
A 55-year-old man complains of exertional chest pain.
Key findings:
- Wide pulse pressure
- Normal coronary angiogram
- Severe AR on echo
Diagnosis: Angina due to reduced diastolic coronary perfusion
Management:
Surgical AVR
6. Marfan Syndrome with AR
Presentation:
A 28-year-old tall male with long limbs presents for evaluation.
Key findings:
- Aortic root dilation
- Moderate AR
- Family history of sudden death
Diagnosis: AR due to aortic root dilation (Marfan syndrome)
Management:
Beta-blocker or ARB, serial imaging, elective surgery if thresholds reached
7. Austin Flint Murmur Case
Presentation:
A 60-year-old man has exertional dyspnea.
Key findings:
- Early diastolic murmur at LSB
- Mid-diastolic rumble at apex
- No opening snap
Diagnosis: Severe AR with Austin Flint murmur
Management:
Evaluate LV size and plan surgery
8. Acute AR with Absent Murmur
Presentation:
A 50-year-old man with acute pulmonary edema.
Key findings:
- Hypotension
- Very faint or absent murmur
- Echo: torrential AR
Diagnosis: Acute severe AR
Management:
Urgent surgery; murmur absent due to pressure equalization
9. Bicuspid Aortic Valve AR
Presentation:
A 32-year-old man with exertional dyspnea.
Key findings:
- Systolic click
- Echo: bicuspid valve, severe AR
- Dilated ascending aorta
Diagnosis: AR due to bicuspid aortic valve
Management:
AVR ± ascending aorta replacement
10. AR with Hypertension
Presentation:
A 65-year-old hypertensive patient with mild dyspnea.
Key findings:
- BP 170/60 mmHg
- Moderate AR on echo
Diagnosis: Chronic AR worsened by uncontrolled hypertension
Management:
ACE inhibitors / ARBs, BP control, echo follow-up
11. AR in Pregnancy
Presentation:
A 30-year-old pregnant woman with known AR presents in second trimester.
Key findings:
- Mild dyspnea
- Echo: moderate AR, preserved EF
Diagnosis: Chronic AR in pregnancy
Management:
Medical management, avoid surgery unless life-threatening
12. AR with Heart Failure
Presentation:
A 60-year-old man presents with orthopnea and PND.
Key findings:
- S3 gallop
- Severe AR
- EF 45%
Diagnosis: Decompensated chronic AR with HFrEF
Management:
Diuretics, vasodilators, urgent AVR
13. Holodiastolic Flow Reversal Case
Presentation:
A 52-year-old asymptomatic patient under follow-up.
Key findings:
- Echo: holodiastolic flow reversal in descending aorta
Diagnosis: Severe AR despite minimal symptoms
Management:
Assess LV dimensions → likely surgery
14. AR with Atrial Fibrillation
Presentation:
A 64-year-old man with palpitations and dyspnea.
Key findings:
- Irregularly irregular pulse
- LA enlargement
- Severe AR
Diagnosis: Chronic AR with AF
Management:
Rate control, anticoagulation, valve surgery
15. Post-Rheumatic AR
Presentation:
A 55-year-old woman with history of rheumatic fever.
Key findings:
- AR murmur
- Associated mitral valve disease
Diagnosis: Rheumatic mixed valve disease with AR
Management:
Combined valve assessment and surgical planning
16. AR with LVESD Index Crossing Threshold
Presentation:
Asymptomatic patient on surveillance.
Key findings:
- Indexed LVESD = 26 mm/m²
- EF preserved
Diagnosis: Severe AR with surgical criteria met
Management:
Elective AVR
17. AR with Syncope
Presentation:
A 58-year-old collapses during exertion.
Key findings:
- Severe AR
- Reduced cerebral perfusion due to low diastolic BP
Diagnosis: Advanced AR with hemodynamic compromise
Management:
Urgent valve replacement
18. AR with Endocarditis Prophylaxis Question
Presentation:
A patient with native AR asks about dental extraction.
Key findings:
- No prior endocarditis
- Native valve only
Diagnosis: Chronic AR without high-risk features
Management:
No antibiotic prophylaxis indicated
19. AR with MRI Confirmation
Presentation:
A patient with discordant echo findings.
Key findings:
- Echo inconclusive
- MRI: regurgitant fraction 55%
Diagnosis: Severe AR confirmed by cardiac MRI
Management:
Surgical referral
20. Delayed Surgery Consequence
Presentation:
A patient delayed AVR despite indications.
Key findings:
- EF now 35%
- Persistent LV dilation post-AVR
Diagnosis: Irreversible LV dysfunction due to delayed surgery
Management:
Heart failure therapy + guarded prognosis