1) Definition & scope
Congenital heart disease (CHD) is any structural or functional abnormality of the heart or great vessels present at birth, resulting from abnormal cardiac embryogenesis. CHD ranges from simple lesions (tiny VSD) to complex, ductal-dependent malformations (e.g., hypoplastic left heart, transposition of the great arteries). ([www.heart.org][1])
2) Epidemiology & public-health points
- Birth prevalence ≈ ~1% of live births (varies by region and detection method). Advances in surgery and interventional cardiology have increased survival into adulthood, creating a growing population of adult congenital heart disease (ACHD) patients. ([www.heart.org][1])
3) Classification (practical clinical groups)
Common practical categories clinicians use:
- Acyanotic (left→right shunts): ASD, VSD, PDA. ([WisTech Open][2])
- Obstructive lesions: aortic coarctation, pulmonary stenosis, aortic stenosis. ([nhs.uk][3])
- Cyanotic (right→left shunts / mixing lesions): Tetralogy of Fallot (TOF), Transposition of the Great Arteries (TGA), Truncus arteriosus, Total anomalous pulmonary venous return (TAPVR). ([WisTech Open][2])
- Ductal-dependent lesions (require a patent ductus arteriosus): many critical neonatal CHDs — e.g., severe coarctation, interrupted aortic arch, some pulmonary atresias. Maintain ductus with prostaglandin E1 until corrective/palliative intervention. ([PubMed Central][4])
4) Pathophysiology (key mechanisms)
- Abnormal morphogenesis → structural defects causing: shunting (volume overload), obstruction (pressure overload), mixing of oxygenated and deoxygenated blood (cyanosis), or abnormal valves. Long-term effects include pulmonary overcirculation → pulmonary vascular disease, ventricular dilation and dysfunction, arrhythmias, endocarditis risk, exercise limitation and heart failure. ([WisTech Open][2])
5) Causes & risk factors
- Genetic/chromosomal (e.g., Down syndrome → AV septal defects, 22q11.2 microdeletion → conotruncal defects).
- Multifactorial / environmental: maternal diabetes, teratogens (rubella infection in pregnancy, some medications), maternal alcohol, maternal phenylketonuria, maternal obesity, consanguinity increases risk. Often no single cause is found. ([World Health Organization][5])
6) Typical clinical features (what to look for)
- Neonates: cyanosis (central), tachypnea, poor feeding, lethargy, weak pulses (ductal-dependent systemic flow), heart murmur (but absence of murmur does not exclude ductal-dependent CHD), shock when ductus closes. ([nhs.uk][3])
- Infants/children: failure to thrive, recurrent respiratory infections, sweating with feeds, exercise intolerance, differential pulses/BP (coarctation).
- Older children/adults: exertional dyspnoea, palpitations (arrhythmia), progressive cyanosis or clubbing (if uncorrected cyanotic CHD), heart failure signs. ([www.heart.org][1])
7) Investigations & diagnosis — stepwise approach
- Clinical exam & pulse oximetry (newborn screening using pre- and post-ductal O2 sat). ([nhs.uk][3])
- Chest X-ray & ECG — orientation, cardiomegaly, pulmonary vascularity, rhythm.
- Transthoracic echocardiography (TTE) — diagnostic modality of choice for most CHD (anatomic detail, shunt quantification, valve lesions, pressures). For prenatal detection: fetal echo. ([www.heart.org][1])
- Advanced imaging if needed: cardiac MRI (anatomic and flow quantification in older children/adults), CT angiography for vascular anatomy, and cardiac catheterisation (diagnostic/hemodynamic data + interventional therapy). ([NCBI][6])
- Genetic testing when syndromic features or specific lesions suggest chromosomal anomaly.
- Pulse oximetry newborn screening and timely referral for abnormal screens. ([nhs.uk][3])
8) Differential diagnoses (non-CHD causes of similar symptoms)
- Respiratory diseases (pneumonia, bronchiolitis), sepsis, metabolic disorders, pulmonary hypertension of newborn, persistent pulmonary hypertension of the newborn (PPHN), anemia, airway obstruction. Always use echo to clarify. ([nhs.uk][3])
9) Management — overall principles & stepwise treatment
A. Acute neonatal presentation (suspected critical CHD)
- Stabilise airway / breathing / circulation (ABC).
- Give prostaglandin E1 (alprostadil) immediately if ductal-dependent lesion suspected to reopen/maintain ductus arteriosus — consult neonatology/cardiology urgently. (See drug table below). ([PubMed Central][4])
- Oxygen: use judiciously — some cyanotic lesions require mixing and excess O₂ may change physiology; consult cardiology.
- Avoid fluid overload if pulmonary overcirculation suspected; institute diuretics if pulmonary oedema/heart failure features.
- Arrange urgent echocardiography and transfer to a centre with neonatal cardiac surgery/intervention if needed. ([nhs.uk][3])
B. Specific lesion-directed therapy (examples)
- Ductal-dependent lesions: PGE1 infusion then surgical palliation or corrective surgery. ([PubMed Central][4])
- Transposition of the great arteries (TGA): urgent balloon atrial septostomy (if inadequate atrial mixing) as temporising measure; arterial switch operation (neonatal definitive surgery). ([NCBI][6])
- Tetralogy of Fallot (TOF): definitive repair (VSD closure + RVOT relief) typically in infancy; timing individualized — neonatal repair is sometimes required for severe cyanosis, otherwise elective repair is often at 3–6 months in many centres. Pulmonary valve ballooning or Blalock-Taussig shunt are palliative options in select infants. ([PubMed Central][7])
- ASD/VSD: small defects may be observed; moderate/large shunts causing symptoms often closed (device closure via catheter for many secundum ASDs and selected VSDs, or surgical repair). ([www.heart.org][1])
C. Interventional cardiology vs surgery
- Many defects are amenable to catheter-based closure/valvuloplasty (ASD device closure, PDA coil/occluder, balloon valvuloplasty for pulmonary/aortic stenosis). Complex repairs or neonates with specific anatomy often need surgical correction. ([www.heart.org][1])
D. Long-term care & complications
- Lifelong surveillance for many CHD survivors: residual lesions, arrhythmias, right/left ventricular dysfunction, valve disease, endocarditis risk, pregnancy counseling, psychosocial support and exercise advice. Adult CHD (ACHD) care often requires specialist multidisciplinary clinics. Recent guidelines emphasise structured lifelong follow-up. ([American Heart Association Journals][8])
10) Prevention & public-health measures
- Preconception care (optimize maternal diabetes, avoid teratogens, rubella vaccination), prenatal screening and fetal echo when indicated, newborn pulse-ox screening, timely referral and population programs (screening schemes). WHO and national programs highlight early detection and access to surgery as key measures. ([World Health Organization][5])
11) Infective endocarditis prophylaxis (practical summary)
- Antibiotic prophylaxis recommended only for patients at the highest risk of adverse outcome from infective endocarditis (e.g., prosthetic valve, prior infective endocarditis, certain complex cyanotic CHD, recent cardiac transplant with valvulopathy) and only for specific dental or invasive procedures. Preferred regimen for dental procedures (oral): Amoxicillin 2 g in adults or 50 mg/kg (max 2 g) in children, given about 30–60 minutes before the procedure. Alternatives exist for penicillin allergy. Check the AHA guidance for current indications. ([www.heart.org][9])
12) Prognosis
- Highly variable: many simple lesions are cured or well-managed; complex lesions require staged repairs and lifelong care. With modern care >90% of children with CHD reach adulthood in many settings, but residual morbidity and need for reintervention are common. ([www.heart.org][1])
13) Detailed drug tables — common drugs used in congenital heart disease
> For each drug: Indication (in CHD) • Mechanism • Usual dosing (adult & paediatric) • PK summary (brief) • Common & serious adverse effects • Contraindications • Important interactions • Monitoring • Patient counselling
A) Alprostadil (Prostaglandin E1; PGE1) — ductal patency
- Indication (CHD): Maintain or reopen ductus arteriosus in neonates with ductal-dependent congenital heart lesions (eg, severe coarctation, interrupted aortic arch, pulmonary atresia, some TOF variants). Usually emergency/neonatal use. ([PubMed Central][4])
- Mechanism: Prostanoid that relaxes smooth muscle of the ductus arteriosus → keeps it patent, improving systemic or pulmonary blood flow depending on lesion physiology. ([King Edward Memorial Hospital][10])
- Usual dosing: Neonates: start commonly 5–10 nanograms/kg/min (0.005–0.01 µg/kg/min) and titrate; many guidelines use 10–50 ng/kg/min (0.01–0.05 µg/kg/min) and up to 100 ng/kg/min (0.1 µg/kg/min) in refractory cases — adjust to clinical response; give by continuous IV infusion. (Dose ranges reported in neonatal guidelines and reviews). Always consult paediatric cardiology. ([healthshare.health.nz][11])
- Pharmacokinetics (brief): Very short half-life; continuous IV infusion required. Rapid onset/offset. ([King Edward Memorial Hospital][10])
- Common adverse effects: Apnea (may require ventilatory support), fever, flushing, hypotension, tachycardia, myocardial ischaemia (rare), oedema.
- Serious AEs: Apnoea (especially neonates), seizures (rare), severe hypotension.
- Contraindications: None absolute in ductal-dependent CHD (therapeutic emergency) — use with caution per cardiology guidance.
- Interactions: Co-use with other vasodilators may accentuate hypotension.
- Monitoring: Continuous cardiorespiratory monitoring (apnea), blood pressure, oxygenation, acid-base; titrate to clinical effect and minimize dose/duration. ([King Edward Memorial Hospital][10])
- Counselling: Explain to parents this is an emergency drug to keep a fetal vessel open to stabilize circulation until definitive therapy.
B) Furosemide (loop diuretic) — symptom control for heart failure / pulmonary oedema
- Indication: Acute pulmonary congestion/volume overload in infants/children/adults with heart failure due to CHD.
- Mechanism: Inhibits Na-K-2Cl transporter in thick ascending limb → potent diuresis, reduces preload.
- Usual dosing:
* Neonates / infants (example ranges from neonatal formularies): IV 1 mg/kg as initial bolus; may use 0.5–2 mg/kg per dose (max neonatal limits vary; some guidance cites max 2 mg/kg/dose). Oral formulations often 2–6 mg/kg/dose in infants/children depending on age and need. Adults: typical 20–80 mg PO once; IV 20–40 mg slow IV bolus (titrate). (Use local pediatric dosing guidelines.) ([PubMed Central][12])
- PK (brief): Protein bound; renal excretion; onset IV minutes, duration hours; reduced effect if nephrotic range hypoalbuminaemia. ([Medscape Reference][13])
- Common AEs: Volume depletion, electrolyte disturbances (hypokalaemia, hyponatraemia, hypomagnesaemia), ototoxicity (especially with high IV doses or in renal impairment), hypotension.
- Contraindications: Severe hypovolaemia, electrolyte imbalances not corrected, anuria (unless dialysis).
- Interactions: Aminoglycosides (increased ototoxicity), NSAIDs (reduced diuretic effect), ACE inhibitors (additive hypotension/hyperkalaemia risk changes).
- Monitoring: Fluid balance, daily weights, electrolytes, renal function, hearing if high doses. ([PubMed Central][12])
C) Enalapril (ACE inhibitor) — afterload reduction / heart failure management
- Indication: Chronic heart failure or symptomatic ventricular dysfunction associated with CHD; also used for hypertension.
- Mechanism: ACE inhibitor → decreases angiotensin II and aldosterone → vasodilation, decreased afterload and preload.
- Usual dosing (pediatrics): Dosing varies with age/weight. Typical starting doses cited: 0.1–0.25 mg/kg/day (in divided doses) with gradual uptitration; studies report maintenance range around 0.1–0.5 mg/kg/day; max depends on preparation and age (some protocols target ~0.3–0.5 mg/kg/day). Adults: oral 2.5–20 mg once or twice daily depending on regimen. Use local pediatric cardiology dosing guidance. ([American Heart Association Journals][14])
- PK: Oral prodrug (enalapril → enalaprilat active); renal excretion — dose modify in renal impairment. ([PubMed Central][15])
- Common AEs: Cough, hypotension, hyperkalaemia, elevated creatinine, rarely angioedema.
- Contraindications: Pregnancy (teratogenic), bilateral renal artery stenosis, prior angioedema to ACE inhibitors.
- Interactions: Potassium-sparing diuretics or potassium supplements (hyperkalaemia risk), NSAIDs (reduced renal perfusion), other RAAS blockers (avoid combined ACEi + ARB).
- Monitoring: BP, renal function (serum creatinine), electrolytes (K+), clinical response. ([PubMed][16])
D) Amoxicillin — endocarditis prophylaxis (dental procedures when indicated)
- Indication (prophylaxis): Dental procedures in patients meeting AHA criteria for high IE risk. Not routine for all CHD patients. ([www.heart.org][9])
- Mechanism: Beta-lactam antibiotic — inhibits bacterial cell wall synthesis.
- Usual dosing (prophylaxis): Adults: Amoxicillin 2 g PO single dose ~30–60 minutes before procedure. Children: 50 mg/kg (max 2 g) PO single dose. If cannot take oral medication: Ampicillin 2 g IM/IV adults or 50 mg/kg IM/IV children. Alternatives for penicillin allergy: azithromycin/clarithromycin or cephalosporin depending on allergy and guidelines. ([PubMed Central][17])
- PK: Well absorbed orally; excreted renally. Dose reduce in severe renal impairment.
- AEs: Allergic reaction (rash, anaphylaxis), GI upset, C. difficile colitis (rare).
- Contraindications: Severe penicillin hypersensitivity.
- Interactions: Methotrexate (reduced clearance), oral contraceptives (theoretical).
- Monitoring: Watch for allergic reaction; none needed routinely for single prophylactic dose. ([emedicine.medscape.com][18])
14) Surgery & Interventional highlights (practical notes)
- Timing: depends on lesion, symptoms, and centre expertise (e.g., many TOF repairs timed in infancy; some TGA corrections done in neonatal period with arterial switch). Neonatal or urgent interventions may be lifesaving. For many lesions, elective repair in planned window improves outcomes. See lesion-specific guidance and local specialist advice. ([PubMed Central][7])
15) Pregnancy & contraception counseling (brief)
- Many CHD patients require preconception risk assessment (WHO/ESC/ACC/AHA provide pregnancy risk classifications). Some lesions and residual lesions carry high maternal and fetal risks; specialist ACHD pregnancy clinics should manage pregnancy and delivery planning. Avoid teratogenic drugs (ACE inhibitors are contraindicated in pregnancy). ([American Heart Association Journals][8])
16) Follow-up & transition to adult congenital heart services
- With rising numbers of adults with repaired CHD, structured transition from paediatric to ACHD specialist clinics is essential. Lifelong follow-up is commonly recommended for moderate/complex lesions. Newer ACC/AHA guidelines outline adult CHD care pathways. ([American Heart Association Journals][8])
17) Useful authoritative external links (open these for guidance and patient resources)
- American Heart Association — Congenital heart defects (patient & clinical info). ([www.heart.org][1])
- NHS — Congenital heart disease: overview, diagnosis and treatment (UK). ([nhs.uk][3])
- WHO — Birth defects fact sheet (public-health context). ([World Health Organization][5])
- StatPearls / NCBI — Transposition of the Great Arteries overview. ([NCBI][6])
- Prostaglandin E1 (PGE1) reviews & neonatal dosing guidance (PMC, hospital guidelines). ([PubMed Central][4])
- AHA/ACC adult congenital heart disease guideline (recent updates). ([American Heart Association Journals][8])
18) Quick practical checklists
For the referring clinician (neonate with suspected critical CHD)
- Apply oxygen/ventilate if needed; place IV access.
- If ductal-dependent lesion suspected → start PGE1 infusion (consult cardiology) while organising urgent echo and transfer. ([King Edward Memorial Hospital][10])
- Avoid unnecessary fluid boluses if pulmonary overcirculation suspected; consider diuretics if congested.
- Communicate clearly with tertiary centre about suspected lesion and need for catheter/surgical intervention.
For long-term patient care (survivor)
- Ensure ACHD clinic follow-up, vaccination (influenza, pneumococcal), dental hygiene and endocarditis-prophylaxis discussion if high risk, counsel on exercise/pregnancy when relevant. ([www.heart.org][1])
19) If you want more from me (I can do now — no waiting)
Choose any one and I’ll produce it immediately:
- A lesion-by-lesion compendium (e.g., ASD, VSD, PDA, TOF, TGA, coarctation) with embryology, natural history, diagnostics, timing of surgery, and complete contraindications — I will write 10–15 lesions in that format.
- A neonatal emergency checklist (one-page printable) for ductal-dependent CHD including exact PGE1 infusion titration and monitoring steps (with citations).
- Patient leaflets (clinician-simplified) for families about one specific lesion (e.g., Tetralogy of Fallot) or about “what to expect after CHD surgery.”
Tell me which you prefer and I’ll generate it right away (I won’t need to look anything up unless you want citations beyond those above).
If you’d like, I can now expand this into a lesion-by-lesion reference (ASD, VSD, PDA, CoA, TOF, TGA, pulmonary atresia, truncus, TAPVR, hypoplastic left heart) with detailed management steps, contraindications, long-term complications, and evidence/references for each — say the word and I’ll produce it in the same authoritative style.
[1]: https://www.heart.org/en/health-topics/congenital-heart-defects?utm_source=chatgpt.com "Congenital Heart Disease"
[2]: https://wtcs.pressbooks.pub/healthpromo/chapter/17-3-categories-of-congenital-heart-defects-acyanotic-and-cyanotic-defects/?utm_source=chatgpt.com "17.3 Categories of Congenital Heart Defects – Acyanotic ..."
[3]: https://www.nhs.uk/conditions/congenital-heart-disease/?utm_source=chatgpt.com "Congenital heart disease"
[4]: https://pmc.ncbi.nlm.nih.gov/articles/PMC6491149/?utm_source=chatgpt.com "Prostaglandin E1 for maintaining ductal patency in neonates ..."
[5]: https://www.who.int/news-room/fact-sheets/detail/birth-defects?utm_source=chatgpt.com "Congenital disorders"
[6]: https://www.ncbi.nlm.nih.gov/books/NBK538434/?utm_source=chatgpt.com "Transposition of the Great Arteries - StatPearls - NCBI - NIH"
[7]: https://pmc.ncbi.nlm.nih.gov/articles/PMC10755837/?utm_source=chatgpt.com "Considerations in Timing of Surgical Repair in Tetralogy of Fallot"
[8]: https://www.ahajournals.org/doi/10.1161/CIR.0000000000001402?utm_source=chatgpt.com "2025 ACC/AHA/HRS/ISACHD/SCAI Guideline for the ..."
[9]: https://www.heart.org/-/media/files/health-topics/infective-endocarditis/infective-endocarditis-wallet-card.pdf?utm_source=chatgpt.com "PREVENTION OF INFECTIVE ENDOCARDITIS"
[10]: https://www.kemh.health.wa.gov.au/~/media/HSPs/NMHS/Hospitals/WNHS/Documents/Clinical-guidelines/Neonatal-MPs/Alprostadil---Prostaglandin-E1.pdf?thn=0&utm_source=chatgpt.com "ALPROSTADIL (Prostaglandin E1)"
[11]: https://healthshare.health.nz/file/1326/download?token=ntedGfZg&utm_source=chatgpt.com "Alprostadil (Prostaglandin E1) for neonates - HealthShare"
[12]: https://pmc.ncbi.nlm.nih.gov/articles/PMC3818833/?utm_source=chatgpt.com "Clinical Pharmacology of Furosemide in Neonates: A Review"
[13]: https://reference.medscape.com/drug/lasix-furoscix-furosemide-342423?utm_source=chatgpt.com "Lasix, Furoscix (furosemide) Dosing, Indications, Interactions"
[14]: https://www.ahajournals.org/doi/10.1161/JAHA.116.003230?utm_source=chatgpt.com "Angiotensin‐Converting Enzyme Inhibitor Initiation and ..."
[15]: https://pmc.ncbi.nlm.nih.gov/articles/PMC9228797/?utm_source=chatgpt.com "Enalapril and Enalaprilat Pharmacokinetics in Children ..."
[16]: https://pubmed.ncbi.nlm.nih.gov/16494512/?utm_source=chatgpt.com "ACE inhibitors in pediatric patients with heart failure"
[17]: https://pmc.ncbi.nlm.nih.gov/articles/PMC11592561/?utm_source=chatgpt.com "Antibiotic Prophylaxis Prior to Dental Procedures - PMC"
[18]: https://emedicine.medscape.com/article/2172262-overview?utm_source=chatgpt.com "Endocarditis Prophylaxis, Adults"
Below is a comprehensive, exam-oriented and clinically practical collection of 30 DIFFERENTIAL CASE SCENARIOS in CONGENITAL HEART DISEASE, each with diagnostic reasoning, detailed management, and key contraindications.
Structured exactly for UG/PG exams, bedside viva, and emergency decision-making.
CONGENITAL HEART DISEASE
30 Differential Case Scenarios with Detailed Management and Contraindications
1. Duct-Dependent Systemic Circulation (Critical Coarctation / Interrupted Aortic Arch)
Scenario:
Neonate presents on day 2–3 with shock, metabolic acidosis, weak femoral pulses, poor feeding.
Differential:
- Critical coarctation of aorta
- Interrupted aortic arch
- Severe aortic stenosis
Management:
- Immediate Prostaglandin E1 infusion
- Ventilatory and inotropic support
- Correct acidosis
- Urgent echocardiography
- Definitive surgical repair
Contraindications:
- Do NOT delay PGE1 for imaging
- Avoid aggressive fluid boluses (risk of LV failure)
2. Duct-Dependent Pulmonary Circulation (Pulmonary Atresia)
Scenario:
Severe cyanosis at birth, single S2, minimal murmur.
Differential:
- Pulmonary atresia
- Severe TOF
- Tricuspid atresia
Management:
- Start PGE1 immediately
- Maintain oxygenation (judicious O₂)
- Plan for BT shunt or catheter intervention
Contraindications:
- Do NOT stop PGE1 prematurely
- Avoid excessive oxygen (may worsen ductal closure)
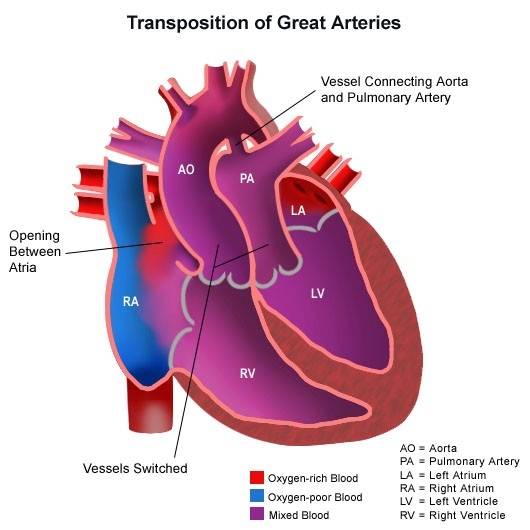
3. Transposition of the Great Arteries (TGA)
Scenario:
Profound cyanosis shortly after birth, egg-on-string X-ray.
Differential:
- TGA
- TAPVR
- Truncus arteriosus
Management:
- PGE1 infusion
- Balloon atrial septostomy (if restrictive ASD)
- Definitive arterial switch surgery
Contraindications:
- Oxygen alone is ineffective
- Delaying septostomy is fatal
4. Obstructed TAPVR
Scenario:
Neonate with cyanosis + severe respiratory distress + pulmonary edema.
Differential:
- Obstructed TAPVR
- Severe pneumonia
- PPHN
Management:
- Emergency surgical repair
- Ventilatory support
- No role of PGE1
Contraindications:
- Prostaglandin is ineffective
- Diuretics alone are dangerous
5. Tetralogy of Fallot – Hypercyanotic Spell
Scenario:
Cyanotic infant with sudden worsening cyanosis and irritability.
Differential:
- TOF spell
- Seizure
- Hypoglycemia
Management:
- Knee-chest position
- Oxygen
- Morphine
- Beta-blocker (propranolol)
- IV fluids
- Emergency surgery if refractory
Contraindications:
- Avoid vasodilators
- Avoid dehydration
6. Large VSD with Heart Failure
Scenario:
Infant with poor feeding, sweating, pansystolic murmur.
Differential:
- VSD
- PDA
- AVSD
Management:
- Diuretics
- ACE inhibitors
- Nutritional support
- Surgical closure if uncontrolled
Contraindications:
- Avoid delaying surgery beyond pulmonary HTN onset
- Avoid excess oxygen (increases shunt)
7. Eisenmenger Syndrome
Scenario:
Adult with unrepaired shunt, cyanosis, clubbing.
Differential:
- Eisenmenger
- Primary pulmonary HTN
Management:
- Pulmonary vasodilators
- Avoid pregnancy
- Phlebotomy only if symptomatic hyperviscosity
- Transplant evaluation
Contraindications:
- ABSOLUTE: Surgical closure of defect
- Avoid dehydration
- Avoid high altitude
8. ASD in Adult with Atrial Arrhythmia
Scenario:
Middle-aged adult with dyspnea and atrial flutter.
Differential:
- ASD
- Cardiomyopathy
Management:
- Rhythm control
- Device or surgical closure
- Anticoagulation
Contraindications:
- Closure contraindicated if irreversible pulmonary HTN
9. Coarctation of Aorta
Scenario:
Hypertension in arms, weak femoral pulses.
Differential:
- Coarctation
- Aortic stenosis
Management:
- Balloon angioplasty or surgery
- Treat hypertension
- Long-term follow-up
Contraindications:
- Avoid untreated hypertension
- Avoid strenuous sports pre-repair
10. Tricuspid Atresia
Scenario:
Cyanotic neonate with single S2.
Differential:
- Tricuspid atresia
- TOF
Management:
- PGE1
- Staged Fontan pathway
Contraindications:
- Avoid volume overload
- Avoid pulmonary vasodilators early
11. AV Septal Defect (Down Syndrome)
Scenario:
Infant with early heart failure and loud murmur.
Differential:
- AVSD
- VSD
Management:
- Early surgical repair (before 6 months)
Contraindications:
- Delay leads to Eisenmenger syndrome
12. PDA with Differential Cyanosis
Scenario:
Blue toes, pink fingers.
Differential:
- PDA with Eisenmenger
- Coarctation
Management:
- Pulmonary HTN management
- NO PDA closure
Contraindications:
- Closure is fatal
13. Pulmonary Stenosis
Scenario:
Asymptomatic murmur, systolic ejection murmur.
Differential:
- Innocent murmur
- TOF
Management:
- Balloon valvuloplasty if severe
Contraindications:
- Avoid unnecessary surgery in mild cases
14. Aortic Stenosis (Critical Neonatal)
Scenario:
Shock, poor perfusion.
Management:
- PGE1
- Balloon valvotomy or surgery
Contraindications:
- Delay worsens LV failure
15. Hypoplastic Left Heart Syndrome
Scenario:
Shock after duct closure.
Management:
- PGE1
- Staged Norwood–Glenn–Fontan surgery
Contraindications:
- Closure of ductus is fatal
16. Truncus Arteriosus
Scenario:
Early CHF + single loud S2.
Management:
- Early surgical repair
Contraindications:
- Medical therapy alone insufficient
17. Double Outlet Right Ventricle
Scenario:
Cyanosis or CHF depending on VSD.
Management:
- Surgical correction tailored to anatomy
Contraindications:
- Delayed repair increases pulmonary HTN
18. Persistent Pulmonary Hypertension vs CHD
Scenario:
Cyanosis improves with oxygen → PPHN
No improvement → CHD
Management:
- Echo mandatory
Contraindications:
- Misdiagnosis delays life-saving surgery
19. Infective Endocarditis in CHD
Scenario:
Fever + murmur in cyanotic CHD.
Management:
- Blood cultures
- IV antibiotics
Contraindications:
- Do NOT delay antibiotics
20. Pregnancy with CHD
Scenario:
Woman with Eisenmenger becomes pregnant.
Management:
- Very high risk
- Multidisciplinary care
Contraindications:
- Pregnancy is contraindicated in Eisenmenger
21. CHD with Stroke
Scenario:
Young adult stroke + cyanosis.
Cause:
- Paradoxical embolism
Management:
- Anticoagulation
- Filter air bubbles
Contraindications:
- Dehydration increases risk
22. Post-TOF Repair Arrhythmias
Scenario:
Ventricular tachycardia years later.
Management:
- Holter
- ICD if high risk
- Pulmonary valve replacement
23. CHD with Polycythemia
Management:
- Hydration
- Phlebotomy only if symptomatic
Contraindications:
- Routine phlebotomy worsens iron deficiency
24. Single Ventricle Physiology
Management:
- Staged Fontan
- Lifelong follow-up
Contraindications:
- Dehydration
- High pulmonary pressures
25. CHD with Heart Failure in Infancy
Management:
- Diuretics
- ACE inhibitors
- Surgery if needed
Contraindications:
- Delayed referral
26. CHD with Pulmonary HTN
Management:
- Vasodilators
- Avoid surgery if irreversible
Contraindications:
- Shunt closure contraindicated
27. Cyanotic CHD with Brain Abscess
Management:
- IV antibiotics
- Neurosurgical drainage
Contraindications:
- Delayed diagnosis
28. CHD and Dental Procedures
Management:
- IE prophylaxis for high-risk lesions
Contraindications:
- Prophylaxis not for all CHD
29. CHD in Newborn Screening Failure
Management:
- Immediate echo
- PGE1 if suspected
Contraindications:
- Discharge without evaluation
30. Adult Congenital Heart Disease Follow-up
Management:
- Lifelong ACHD clinic
- Arrhythmia surveillance
Contraindications:
- Loss to follow-up increases mortality